Online Database of Chemicals from Around the World
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| SynQuest Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.synquestlabs.com | |||
 | +1 (386) 462-0788 | |||
 | +1 (386) 462-7097 | |||
 | Sales@synquestlabs.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Xuchang Chenhe Bio-pharmaceutical Tech. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.the-chem.com | |||
 | +86 (374) 579-3128 +86 15836530076 | |||
 | +86 (374) 579-3128 | |||
 | wxh5168@126.com wxh5168@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic hydrocarbon reagent |
|---|---|
| Name | 2,3-Dichloro-6-methoxyquinoxaline |
| Molecular Structure | 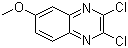 |
| Molecular Formula | C9H6Cl2N2O |
| Molecular Weight | 229.06 |
| CAS Registry Number | 39267-04-4 |
| EC Number | 806-741-3 |
| SMILES | COC1=CC2=C(C=C1)N=C(C(=N2)Cl)Cl |
| Density | 1.5±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 312.6±37.0 °C 760 mmHg (Calc.)* |
| Flash point | 142.9±26.5 °C (Calc.)* |
| Index of refraction | 1.638 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2,3‑Dichloro‑6‑methoxyquinoxaline is a halogenated quinoxaline derivative in which chlorine atoms occupy the 2‑ and 3‑positions of the quinoxaline ring and a methoxy group is present at the 6‑position. The quinoxaline core (a bicyclic system comprised of a benzene ring fused to a pyrazine ring) is a common scaffold in medicinal chemistry due to its planar aromatic nature and ability to engage in π-π interactions, hydrogen bonding, and other non‑covalent interactions. The dichloro substitution makes the molecule particularly reactive in nucleophilic aromatic substitution (SNAr) chemistry, while the methoxy substituent modulates the electron density of the ring and can influence both reactivity and solubility. In synthesis, one plausible route to 2,3-dichloroquinoxalines involves the reaction of quinoxaline-2,3(1H,4H)-dione with phosphoryl chloride (POCl₃) under reflux, converting hydroxyl or keto functionalities at the 2 and 3 positions into chloro substituents. Following this, the methoxy group could be introduced via electrophilic substitution on the benzene portion of the quinoxaline ring—though detailed published procedures for specifically 6-methoxy substitution are limited. Applications of substituted dichloroquinoxalines include their use as intermediates in the synthesis of biologically active compounds. Quinoxaline derivatives are found in pharmaceuticals, agrochemicals, and functional materials, acting as building blocks for further elaboration. The dichloro functionality allows coupling or substitution to generate derivatives with tailored properties, while the methoxy group might enhance lipophilicity or adjust electronic properties, influencing binding to biological targets. In practical handling, 2,3‑dichloro‑6‑methoxyquinoxaline is likely to be handled under dry conditions because of the sensitivity of chloroquinoxalines to hydrolysis or decomposition. It would dissolve in common organic solvents and can be purified by standard methods (recrystallization or chromatography) after synthesis or substitution reactions. Because there is a lack of peer‑reviewed articles specifically on 2,3-dichloro-6-methoxyquinoxaline, it is primarily documented in chemical catalogues and databases as a synthetic intermediate rather than as a final active compound. References Synfacts (2019) Synthesis of Grazoprevir. Synfacts 15 01 DOI: 10.1055/s-0037-1612028 Synfacts (2013) Synthesis of MK-5172. Synfacts 09 10 DOI: 10.1055/s-0033-1339864 Chemistry of Heterocyclic Compounds (1976) Investigation of heterocyclic quinones. Chemistry of Heterocyclic Compounds 12 1767–1772 DOI: 10.1007/bf00476731 |
| Market Analysis Reports |