Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| GLSyntech, LLC | USA | |||
|---|---|---|---|---|
 | www.glsyntech.com | |||
 | +1 (215) 367-5129 | |||
 | +1 (215) 701-4982 | |||
 | sales@glsyntech.com | |||
| Chemical manufacturer | ||||
| BetaPharma Inc. | USA | |||
|---|---|---|---|---|
 | www.betapharma.com | |||
 | +1 (203) 315-5062 | |||
 | sales@betapharma.com | |||
| Chemical manufacturer since 1996 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Ukrorgsyntez Ltd. | Ukraine | |||
|---|---|---|---|---|
 | www.uorsy.com | |||
 | +38 (044) 531-9497 | |||
 | +38 (044) 531-9497 | |||
 | sales.eu@uorsy.com sales.us@uorsy.com | |||
| Chemical manufacturer since 2001 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Carboxylic acid |
|---|---|
| Name | 5-Fluoro-2-methoxybenzoic acid |
| Molecular Structure | 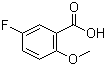 |
| Molecular Formula | C8H7FO3 |
| Molecular Weight | 170.14 |
| CAS Registry Number | 394-04-7 |
| EC Number | 627-722-4 |
| SMILES | COC1=C(C=C(C=C1)F)C(=O)O |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 87 - 91 °C (Expl.) |
| Boiling point | 294.0±20.0 °C 760 mmHg (Calc.)* |
| Flash point | 131.6±21.8 °C (Calc.)* |
| Index of refraction | 1.524 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
5-Fluoro-2-methoxybenzoic acid is a chemical compound that belongs to the family of substituted benzoic acids. It is characterized by a benzoic acid backbone with a methoxy group (-OCH₃) at the 2-position and a fluorine atom at the 5-position. This molecule is of significant interest in the fields of organic synthesis, medicinal chemistry, and materials science due to its structural properties and potential applications. The discovery of 5-fluoro-2-methoxybenzoic acid stems from the exploration of halogenated and methoxy-substituted aromatic acids, which have become a major focus in the development of new pharmaceutical compounds and materials. The introduction of the fluorine atom in the 5-position imparts unique electronic properties to the molecule, such as increased acidity and enhanced lipophilicity, making it suitable for various chemical transformations and biological applications. The primary method for synthesizing 5-fluoro-2-methoxybenzoic acid involves the functionalization of benzoic acid derivatives. The fluorine atom is typically introduced through electrophilic aromatic substitution reactions using fluorinating agents, such as fluorine gas or reagents like Selectfluor. The methoxy group is introduced at the 2-position using standard methylation techniques, often employing reagents such as dimethyl sulfate or methyl iodide in the presence of a base like potassium carbonate. These reactions are well-established and allow for the efficient production of 5-fluoro-2-methoxybenzoic acid. In terms of its applications, 5-fluoro-2-methoxybenzoic acid is widely used in the synthesis of pharmaceuticals, particularly in the development of anti-inflammatory, analgesic, and anticancer agents. The methoxy group enhances the compound’s ability to penetrate biological membranes, while the fluorine atom may influence its pharmacokinetic properties, such as stability, bioavailability, and receptor binding. As such, derivatives of this compound are often studied for their potential therapeutic properties, particularly as nonsteroidal anti-inflammatory drugs (NSAIDs) or as part of drug development programs targeting various diseases. Beyond its role in medicinal chemistry, 5-fluoro-2-methoxybenzoic acid is also used in materials science. It can serve as a precursor to the synthesis of polyfunctional materials, including polymers and liquid crystals. The introduction of fluorine and methoxy groups into the aromatic ring can significantly modify the electronic and optical properties of the resulting materials, making them useful in the development of electronic devices, coatings, and sensors. Another area where 5-fluoro-2-methoxybenzoic acid shows promise is in the field of agrochemicals. Its derivatives have been explored for their potential as herbicides or fungicides, where the combination of fluorine and methoxy groups can lead to enhanced biological activity and selective toxicity toward target organisms. The compound also finds use as an intermediate in the synthesis of other fine chemicals. Due to its functional groups, 5-fluoro-2-methoxybenzoic acid can undergo various chemical transformations, such as esterification, amidation, or coupling reactions, to produce a wide array of compounds. Its reactivity makes it a versatile building block in organic chemistry, particularly in the preparation of more complex aromatic systems. In conclusion, 5-fluoro-2-methoxybenzoic acid is an important compound in both organic synthesis and medicinal chemistry. Its structure, featuring both a fluorine atom and a methoxy group on a benzoic acid backbone, imparts unique properties that make it valuable in the development of pharmaceuticals, agrochemicals, and advanced materials. Its synthetic accessibility and reactivity further enhance its utility in the design of new molecules with diverse applications, ensuring its continued relevance in research and industrial applications. References 2010. Synthesis and antimicrobial activity of cyclopentyl-substituted halogenophenoxyacetic acids and their amides. Pharmaceutical Chemistry Journal, 44(9). DOI: 10.1007/s11094-010-0482-x 2018. One-Step O-Demethyl-Decarboxylation of ortho-Methoxy Aromatic Carboxylic Acids. Synthesis, 50(10). DOI: 10.1055/s-0036-1591912 2006. Halogen Directed ortho-Lithiation Groups. Science of Synthesis, 1. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-008-00229 |
| Market Analysis Reports |