Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Qingdao Kingway Pharmtech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingwaypharm.com | |||
 | +86 (532) 8711-8899 | |||
 | +86 (532) 8411-7788 | |||
 | sales@kingwaypharm.com kingwaypharm@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Ningbo Gino Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ginochem.cn | |||
 | +86 (574) 2788-1160 | |||
 | +86 (574) 2788-1160 | |||
 | info@chem.la | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Tengxiang Imp&Exp Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jinxiangchem.com | |||
 | +86 (25) 8681-9862 +86 13913839793 | |||
 | +86 (25) 8681-9859 | |||
 | sales@jinchemical.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Kunshan Kunshi Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fafachem.com | |||
 | +86 15250298089 | |||
 | 1753428355@qq.com | |||
 | QQ Chat | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Nanjing Shunxiang Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunxiangchem.com | |||
 | +86 (025) 8681-9868 | |||
 | +86 (025) 8681-9859 | |||
 | sales@jinchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Dhruvtara Chemicals Pvt Ltd. | India | |||
|---|---|---|---|---|
 | www.dhruvtarachemicals.com | |||
 | +91 (40) 2715-4607 | |||
 | +91 (40) 2715-4607 | |||
 | info@dhruvtarachemicals.com | |||
| Chemical manufacturer | ||||
| DeLong Chemicals America | USA | |||
|---|---|---|---|---|
 | www.delongchemicals.com | |||
 | +1 (203) 271-9017 | |||
 | sales@delongchemicals.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | Pyridinium tribromide |
| Synonyms | Pyridinium bromide perbromide; Pyridine hydrobromide compound with bromine |
| Molecular Structure | 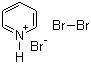 |
| Molecular Formula | C5H6Br3N |
| Molecular Weight | 319.82 |
| CAS Registry Number | 39416-48-3 |
| EC Number | 254-446-8 |
| SMILES | C1=CC=[NH+]C=C1.[Br-].BrBr |
| Melting point | 127-133 °C |
|---|---|
| Water solubility | decomposes |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P264-P270-P280-P301+P317-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P321-P330-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
Pyridinium tribromide is a crystalline ionic compound composed of a pyridinium cation (C5H5NH+) and a tribromide anion (Br3−). It is widely used in organic chemistry as a source of electrophilic bromine (Br2) for bromination reactions. The tribromide anion is a linear, three-bromine species that acts as a stable, solid form of molecular bromine, which is otherwise a volatile and corrosive liquid at room temperature. The compound is typically synthesized by treating pyridine with an excess of bromine. In this reaction, pyridine acts as a base, becoming protonated to form the pyridinium ion, while the bromide ion generated reacts with molecular bromine to form the tribromide ion. The resulting pyridinium tribromide is a red-orange crystalline solid, which is more convenient and safer to handle than elemental bromine. Pyridinium tribromide serves primarily as a brominating agent in laboratory-scale organic synthesis. It is used to brominate a wide variety of organic substrates, including alkenes, alkynes, aromatic compounds, and ketones. In electrophilic aromatic substitution reactions, it acts as a controlled source of bromine, allowing for mono- or polybromination under mild conditions. For example, it is used in the bromination of phenols, anilines, and other activated aromatic rings. In addition to simple halogenation reactions, pyridinium tribromide is also employed in oxidative transformations and deprotection reactions. It has been used in the synthesis of heterocycles and in the preparation of brominated intermediates for pharmaceuticals and agrochemicals. Its selectivity and reactivity can often be tuned by the use of specific solvents or reaction conditions. The advantages of pyridinium tribromide over molecular bromine include its stability in solid form, ease of weighing and handling, and reduced risk of exposure to toxic bromine vapors. It also allows for more precise stoichiometric control in reactions, which is particularly important in small-scale and academic research settings. Pyridinium tribromide is soluble in polar solvents such as acetic acid, ethanol, and dimethylformamide, and it reacts readily upon dissolution, releasing bromine for chemical reactions. In aqueous solution, the tribromide ion exists in equilibrium with bromine and bromide, depending on the concentration and pH. The compound is generally stable at room temperature when stored in a tightly sealed container protected from moisture and light. However, it is sensitive to heat and may decompose at elevated temperatures, releasing bromine gas. Appropriate safety precautions should be taken when handling the compound, including the use of gloves and working in a well-ventilated area or fume hood. Analytical techniques such as nuclear magnetic resonance (NMR) spectroscopy, infrared (IR) spectroscopy, and X-ray crystallography have been used to characterize pyridinium tribromide and confirm the presence of the tribromide anion and the pyridinium cation. The linear structure of the Br3− ion has been confirmed by crystallographic studies. In summary, pyridinium tribromide is a valuable brominating reagent in organic chemistry. It provides a stable, solid alternative to liquid bromine, with broad utility in halogenation and oxidation reactions. Its ease of handling, reliability, and effectiveness make it an important reagent in synthetic laboratories. References 2008. A facile and convenient method for the conversion of thioamides into amides using pyridinium hydrobromide perbromide. Monatshefte für Chemie - Chemical Monthly, 139(4). DOI: 10.1007/s00706-007-0787-y 2007. Kinetics and mechanistic study of pyridinium hydrobromide perbromide-induced carbon-carbon bond scission. Transition Metal Chemistry, 32(3). DOI: 10.1007/s11243-007-0187-5 2008. Synthesis and characterization of 5-bromo-3-sec-butyl-6-methyluracil. Frontiers of Chemistry in China, 3(2). DOI: 10.1007/s11458-008-0029-9 |
| Market Analysis Reports |