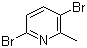
2,5-Dibromo-6-methylpyridine is a halogenated heteroaromatic compound belonging to the class of substituted pyridines. Its molecular formula is C6H5Br2N, and it features a pyridine ring substituted with two bromine atoms at the 2- and 5-positions and a methyl group at the 6-position. The presence of both electron-withdrawing (bromo) and electron-donating (methyl) substituents on the pyridine ring gives the molecule distinct chemical reactivity and synthetic value.
The compound is typically synthesized through stepwise halogenation of 6-methylpyridine (also known as 2-picoline). Bromination reactions using reagents such as bromine (Br2) or N-bromosuccinimide (NBS) under controlled conditions lead to substitution on the aromatic ring. The introduction of bromine atoms occurs selectively, influenced by the directing effects of the nitrogen atom and methyl group. The 2- and 5-positions are particularly susceptible to electrophilic attack due to resonance and inductive effects from the ring substituents.
2,5-Dibromo-6-methylpyridine serves as a useful intermediate in organic synthesis, especially in the preparation of more complex pyridine derivatives. The bromo groups enable palladium-catalyzed cross-coupling reactions such as Suzuki, Sonogashira, and Buchwald–Hartwig reactions, which are essential tools in constructing C–C or C–N bonds. This allows for the formation of various biaryl systems, alkynylpyridines, and amino-substituted pyridines, depending on the choice of coupling partners.
The methyl group at the 6-position provides an additional handle for functionalization, such as oxidation to a carboxylic acid or formylation. This further expands the utility of the compound in the development of pharmaceuticals, agrochemicals, and materials.
In medicinal chemistry, halogenated pyridines like 2,5-dibromo-6-methylpyridine are of interest due to their role as core scaffolds in the design of bioactive molecules. Pyridine rings are commonly found in kinase inhibitors, antimicrobial agents, and central nervous system (CNS)-active compounds. The electron density and steric properties imparted by the bromo substituents can influence binding affinity and selectivity toward specific biological targets.
The compound also finds application in the development of ligands for coordination chemistry. Substituted pyridines can act as nitrogen donors in metal complexes, and the bromo groups may be retained or replaced to tune the electronic properties of the ligands. Such complexes are explored for their catalytic, optical, or magnetic properties.
Due to its reactivity and versatility, 2,5-dibromo-6-methylpyridine is frequently employed in combinatorial chemistry and fragment-based drug discovery, where it serves as a modular unit that can be diversified through chemical transformations. In polymer and materials science, pyridine derivatives have been used as monomers or functional additives for the synthesis of conductive, fluorescent, or coordination-responsive materials.
In summary, 2,5-dibromo-6-methylpyridine is a valuable synthetic intermediate that offers diverse opportunities for derivatization and application across multiple areas of chemical research and development, including pharmaceuticals, catalysis, and advanced materials.
References
2021. Synthesis of BMS-986278. Synfacts, 18(02).
DOI: 10.1055/s-0041-1737176
|





































 GHS05;GHS06 Danger Details
GHS05;GHS06 Danger Details