Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Gelest, Inc. | USA | |||
|---|---|---|---|---|
 | www.gelest.com | |||
 | +1 (215) 547-1015 | |||
 | +1 (215) 547-2484 | |||
 | info@gelest.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Cyanate ester / isocyanate |
|---|---|
| Name | Tris(4-isocyanatophenyl) thiophosphate |
| Molecular Structure | 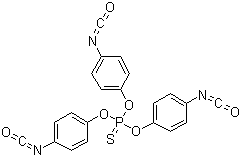 |
| Molecular Formula | C21H12N3O6PS |
| Molecular Weight | 465.38 |
| CAS Registry Number | 4151-51-3 |
| EC Number | 223-981-9 |
| SMILES | C1=CC(=CC=C1N=C=O)OP(=S)(OC2=CC=C(C=C2)N=C=O)OC3=CC=C(C=C3)N=C=O |
| Solubility | 0.000104 mg/L (25 °C water) |
|---|---|
| Density | 1.4±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.637, Calc.* |
| Melting point | 90.27 °C |
| Boiling Point | 561.9±60.0 °C (760 mmHg), Calc.*, 480.00 °C |
| Flash Point | 293.7±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H334 Details | ||||||||||||
| Safety Statements | P233-P260-P264-P270-P271-P284-P301+P317-P304+P340-P330-P342+P316-P403-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Tris(4-isocyanatophenyl) thiophosphate is a compound that combines the functionalities of isocyanate and thiophosphate groups, making it a significant molecule in the field of polymer science and materials chemistry. Its discovery can be attributed to the ongoing search for effective flame retardants and stabilizers for polymers. Researchers were particularly interested in enhancing the thermal and oxidative stability of polymeric materials to meet the increasing demands for safety and performance in various applications. The synthesis of Tris(4-isocyanatophenyl) thiophosphate involves a multi-step chemical process that includes the reaction of isocyanates with thiophosphate esters. The resultant compound features three isocyanate groups attached to a phenyl ring, combined with a thiophosphate moiety, providing a unique chemical structure that imparts specific properties to the material. The presence of isocyanate groups enables the formation of polyurethanes and other polymers through crosslinking reactions, while the thiophosphate group contributes to flame retardancy and improved thermal stability. One of the primary applications of Tris(4-isocyanatophenyl) thiophosphate is as a flame retardant additive in polymer formulations. The compound’s ability to inhibit combustion processes makes it particularly valuable in the production of plastics, foams, and coatings used in construction, automotive, and electrical applications. By incorporating this compound into materials, manufacturers can enhance the safety profile of their products, ensuring compliance with stringent fire safety regulations. In addition to its role as a flame retardant, Tris(4-isocyanatophenyl) thiophosphate serves as a curing agent for polyurethane systems. The isocyanate groups can react with polyols to form crosslinked networks, resulting in durable and heat-resistant materials. This application is particularly important in industries such as automotive and aerospace, where high-performance materials are essential for safety and reliability. Moreover, this compound has been explored for its potential in producing specialty coatings and adhesives. The unique combination of isocyanate and thiophosphate functionalities allows for the development of coatings with enhanced adhesion, flexibility, and resistance to environmental factors. These properties are crucial for applications where durability and longevity are required, such as protective coatings for metal surfaces and automotive finishes. Safety considerations are paramount when working with Tris(4-isocyanatophenyl) thiophosphate due to the presence of isocyanate groups, which can pose health risks if not handled properly. It is essential for manufacturers and users to follow appropriate safety protocols, including using personal protective equipment and ensuring adequate ventilation during handling and processing. Regulatory guidelines must be adhered to in order to mitigate potential hazards associated with its use. In conclusion, Tris(4-isocyanatophenyl) thiophosphate is a versatile compound with significant applications in flame retardancy, polyurethane production, and specialty coatings. Its unique chemical structure enables it to enhance the performance and safety of various materials used across multiple industries. As research continues to evolve, the relevance of this compound in developing safer and more durable products is expected to increase, reflecting the ongoing demand for innovative solutions in materials science. References 1991. The application of size exclusion chromatography equipped with RI and LALLS detectors to study network formation. Polymer Bulletin, 27(3). DOI: 10.1007/bf00294533 2004. Plastics and Composites from Soybean Oil. Natural Fibers, Plastics and Composites, (No volume/issue available). DOI: 10.1007/978-1-4419-9050-1_11 2019. The Development of Sealing Materials Using Modified Epoxyurethane Oligomers. Polymer Science, Series D, 12(4). DOI: 10.1134/s1995421219040129 |
| Market Analysis Reports |