Online Database of Chemicals from Around the World
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Deuterated reagent |
|---|---|
| Name | Aniline-d5 |
| Synonyms | 2,3,4,5,6-pentadeuterioaniline |
| Molecular Structure | 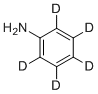 |
| Molecular Formula | C6H2D5N |
| Molecular Weight | 98.16 |
| CAS Registry Number | 4165-61-1 |
| EC Number | 224-015-9 |
| SMILES | [2H]C1=C(C(=C(C(=C1[2H])[2H])N)[2H])[2H] |
| Solubility | 2.082e+004 mg/L (25 °C water) |
|---|---|
| Density | 1.0±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.579, Calc.* |
| Melting point | -6.16 °C |
| Boiling Point | 184.4±0.0 °C (760 mmHg), Calc.*, 183.99 °C |
| Flash Point | 70.0±0.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS05;GHS06;GHS08;GHS09 Danger Details GHS05;GHS06;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301+H311+H331-H317-H318-H341-H351-H372-H410 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P273-P280-P301+P310-P302+P352+P312-P304+P340+P311-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
Aniline-d5 is a deuterated form of aniline, where five hydrogen atoms in the phenyl ring are replaced by deuterium, a stable isotope of hydrogen. The discovery of deuterated compounds like aniline-d5 is linked to advancements in isotope chemistry, particularly in nuclear magnetic resonance (NMR) spectroscopy. The development of deuterated aniline has provided scientists with a valuable tool for studying chemical reactions, molecular structures, and interactions at a detailed level. The synthesis of aniline-d5 involves the reaction of deuterated reagents with benzene derivatives. One common method includes the exchange of hydrogen atoms with deuterium through catalytic deuteration of benzene, followed by subsequent functionalization to introduce the amine group. The deuterium atoms in aniline-d5 are stable, making it ideal for use in spectroscopic applications without concerns about isotopic exchange. The primary application of aniline-d5 is in NMR spectroscopy, particularly as an internal standard or solvent in studies involving organic molecules. The presence of deuterium atoms, which exhibit different magnetic properties than hydrogen, makes aniline-d5 extremely useful for obtaining clearer, more detailed spectra. In proton NMR, the deuterium atoms do not contribute to the proton signals, allowing scientists to isolate and analyze the signals of interest with minimal interference. This feature is crucial when studying complex organic reactions or molecular structures where precise identification of chemical environments is necessary. In addition to its use in NMR spectroscopy, aniline-d5 is also utilized in mass spectrometry. Deuterated compounds are commonly employed as internal standards for quantification due to their similar chemical properties to their non-deuterated counterparts but with a distinct mass shift. This allows for accurate determination of concentrations and improved analytical performance in various studies, including pharmaceutical research, environmental monitoring, and biochemical assays. Aniline-d5 also plays a role in kinetic isotope effect studies, where the difference in reaction rates between deuterium and hydrogen is used to gain insights into reaction mechanisms. By substituting hydrogen atoms with deuterium in aniline-d5, researchers can observe how the bond strength and reaction pathways change, contributing to a deeper understanding of chemical reactivity and catalysis. Furthermore, the development of aniline-d5 and other deuterated compounds has opened new opportunities in the pharmaceutical industry. Deuterium-substituted drugs, often referred to as "heavy drugs," can exhibit improved pharmacokinetic properties, such as slower metabolism and prolonged efficacy. Although aniline-d5 itself is not directly used in drug formulations, its deuterium-labeled structure provides valuable insights for designing such compounds. Overall, the discovery of aniline-d5 has had a significant impact on chemical analysis, reaction mechanism studies, and isotope chemistry. Its applications in NMR spectroscopy, mass spectrometry, and kinetic studies continue to make it an indispensable tool in both academic and industrial research. References 2018. A sensitive method using SPME pre-concentration for the quantification of aromatic amines in indoor air. Analytical and Bioanalytical Chemistry, 410(6). DOI: 10.1007/s00216-018-0862-8 2017. Optimization of Analytical Conditions for a Rapid Determination of Aniline in Environmental Water by Liquid Chromatography/Tandem Mass Spectrometry. Analytical Sciences, 33(10). DOI: 10.2116/analsci.33.1189 2008. Synthesis of Deuterium-Labeled Azo Dyes of the Sudan Family. Synthesis, 2008(4). DOI: 10.1055/s-2008-1032036 |
| Market Analysis Reports |