Online Database of Chemicals from Around the World
| Jiangsu Vcare Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.vcarepharmatech.com | |||
 | +86 (25) 5874-1518 | |||
 | +86 (25) 5874-4115 | |||
 | sales@vcarepharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Qinba Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shangfluoro.com | |||
 | +86 (21) 6517-0832 +86 17301729617 | |||
 | +86 (21) 6517-0832 | |||
 | shangfluoro@hotmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Binlaichem Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.binlaichem.com | |||
 | +86 15963310191 | |||
 | sales@binlaichem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2022 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Ramidus AB | Sweden | |||
|---|---|---|---|---|
 | www.ramidus.se | |||
 | +46 (46) 286-2850 | |||
 | +46 (46) 286-2851 | |||
 | info@ramidus.se | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Tosylate |
|---|---|
| Name | 2,2,2-Trifluoroethyl 4-methylbenzenesulfonate |
| Synonyms | 2,2,2-Trifluoroethyl p-toluenesulfonate |
| Molecular Structure | 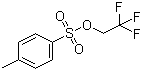 |
| Molecular Formula | C9H9F3O3S |
| Molecular Weight | 254.23 |
| CAS Registry Number | 433-06-7 |
| EC Number | 207-085-5 |
| SMILES | CC1=CC=C(C=C1)S(=O)(=O)OCC(F)(F)F |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 38 - 40 °C (Expl.) |
| Boiling point | 325.3 °C 760 mmHg (Calc.)*, 382.6 - 391.4 °C (Expl.) |
| Flash point | 144.0±26.5 °C (Calc.)*, 113 °C (Expl.) |
| Index of refraction | 1.464 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2,2,2-Trifluoroethyl 4-methylbenzenesulfonate is a fluorinated sulfonate ester with the molecular formula CF3CH2OSO2C6H4CH3. It consists of a trifluoroethyl group linked to a 4-methylbenzenesulfonyl moiety, combining the electron-withdrawing properties of the trifluoromethyl group with the good leaving group ability of the tosylate. The compound is typically a colorless to pale yellow liquid and is highly reactive in nucleophilic substitution reactions due to the sulfonate group, which makes it a versatile intermediate in organic synthesis. The discovery of 2,2,2-trifluoroethyl 4-methylbenzenesulfonate is associated with the broader development of organofluorine chemistry in the 20th century, when chemists explored fluorinated sulfonates as intermediates for incorporating trifluoroethyl groups into organic molecules. The trifluoroethyl moiety is valued for its electron-withdrawing effect, which can enhance the chemical and metabolic stability of downstream products. The combination of the trifluoroethyl group with the tosylate leaving group makes this compound particularly useful for the selective introduction of trifluoroethyl groups via nucleophilic displacement reactions. In organic synthesis, 2,2,2-trifluoroethyl 4-methylbenzenesulfonate is employed as an alkylating agent for nucleophiles such as alcohols, amines, thiols, and carbanions. The sulfonate group is an excellent leaving group, allowing the trifluoroethyl group to be transferred efficiently under mild conditions. This makes the compound a valuable building block for the preparation of trifluoroethylated ethers, amines, thioethers, and other functionalized molecules. Its reactivity is highly useful in medicinal chemistry and agrochemical research, where trifluoroethyl groups can improve lipophilicity, metabolic stability, and biological activity. In pharmaceutical chemistry, the compound is used to synthesize trifluoroethylated intermediates for drug candidates. Incorporation of the trifluoroethyl group into bioactive molecules can modify pharmacokinetic properties, enhance membrane permeability, and improve resistance to metabolic degradation. This makes it valuable for the preparation of enzyme inhibitors, antiviral agents, central nervous system drugs, and other therapeutically relevant compounds. In materials science and specialty chemical synthesis, 2,2,2-trifluoroethyl 4-methylbenzenesulfonate has been used to introduce trifluoroethyl groups into polymers, resins, and functionalized surfaces. Fluorination imparts chemical resistance, hydrophobicity, and thermal stability, enhancing the performance and durability of materials. The tosylate functionality allows for selective reactions that introduce fluorinated moieties without affecting other sensitive functional groups in complex molecules. Analytical chemistry also benefits from trifluoroethyl sulfonates. They can be used as derivatizing agents to improve the volatility and detectability of polar compounds in gas chromatography and mass spectrometry. The trifluoroethyl group enhances detection sensitivity and the overall stability of the derivatives. The development and use of 2,2,2-trifluoroethyl 4-methylbenzenesulfonate highlight its role as a versatile fluorinated reagent. Its combination of a reactive leaving group and a trifluoroethyl moiety makes it a valuable intermediate for organic synthesis, supporting applications in pharmaceuticals, agrochemicals, materials science, and analytical chemistry. The compound exemplifies how selective fluorination and functionalization can expand the utility of sulfonate esters in modern chemical research and industrial processes. References 2011. Radiosynthesis of the tumor hypoxia marker [18F]TFMISO via O-[18F]trifluoroethylation reveals a striking difference between trifluoroethyl tosylate and iodide in regiochemical reactivity toward oxygen nucleophiles. Bioorganic & Medicinal Chemistry, 19(7). DOI: 10.1016/j.bmc.2011.02.026 2010. 2,2,2-Trifluoro-ethyl 4-methyl-benzene-sulfonate. Acta Crystallographica Section E: Structure Reports Online, 66(11). DOI: 10.1107/s1600536810038894 2009. Concise synthesis of halogenated chrysenes ([4]phenacenes) that favor pi-stack packing in single crystals. Organic Letters, 11(15). DOI: 10.1021/ol901693y |
| Market Analysis Reports |