Online Database of Chemicals from Around the World
| ABA Chemicals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.abachem.com | |||
 | +86 (21) 5115-9199 | |||
 | +86 (21) 5115-9188 | |||
 | rimondlong@abachem.com sales@abachem.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhejiang Unipharma-Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.unipharma-chem.com | |||
 | +86 (571) 8580-3833 | |||
 | +86 (571) 8580-3855 | |||
 | info@unipharma-chem.com chenlinfang@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shandong Huimeng Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huimengchem.com | |||
 | +86 (530) 884-2888 +86 18523002917 | |||
 | +86 (530) 884-2666 | |||
 | cindy@szgoldenunion.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Mayue Longteng Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lantu-tech.com | |||
 | +86 15171477138 | |||
 | helen.yan@lantu-tech.com | |||
 | QQ Chat | |||
 | WeChat: 15171477138 | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyran compound |
|---|---|
| Name | 1H-2-Benzopyran-3(4H)-one |
| Synonyms | 2-Hydroxymethylphenylacetic acid lactone; 3,6-Dihydro-4,5-benzo-2-pyrone |
| Molecular Structure | 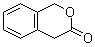 |
| Molecular Formula | C9H8O2 |
| Molecular Weight | 148.16 |
| CAS Registry Number | 4385-35-7 |
| EC Number | 224-493-9 |
| SMILES | C1C2=CC=CC=C2COC1=O |
| Melting point | 83 °C |
|---|---|
| Boiling point | 130 °C (1 mmHg) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
1H-2-Benzopyran-3(4H)-one, commonly known as chromanone or simply coumarin, is a naturally occurring organic compound with the molecular formula C9H6O2. The compound is characterized by a benzene ring fused to a pyrone ring, which is a six-membered oxygen-containing heterocycle. Coumarin is known for its sweet aroma and is found in various plants, especially tonka beans, sweet clover, and some grasses. The discovery of coumarin dates back to the 19th century, when French scientist A. Vogel first isolated coumarin from tonka beans in 1820. Its pleasant vanilla smell quickly made it popular in the perfume industry. Later, the structure of coumarin was elucidated through chemical degradation studies, which led to the development of a more efficient synthetic method for its production. The application of coumarin involves multiple industries, mainly due to its aromatic properties and biological activity. In the flavor and fragrance industry, coumarin is widely used as a flavoring agent in food and a flavoring ingredient in perfumes and cosmetics. Its sweet taste makes it an attractive additive, although its use is regulated due to potential toxicity at high doses. In the pharmaceutical field, coumarin and its derivatives have attracted great interest due to their various biological activities. Coumarin itself has anticoagulant properties and is the basis for the synthesis of several anticoagulant drugs, such as warfarin and acenocoumarol. These drugs inhibit vitamin K epoxide reductase, an enzyme essential for the synthesis of coagulation factors, thereby preventing blood clots in patients at risk for thrombosis and embolism. In addition to anticoagulants, coumarin derivatives also exhibit various pharmacological activities, including anti-inflammatory, antibacterial, antiviral, and anticancer effects. For example, the natural anticoagulant dicoumarol was one of the first coumarin derivatives to be discovered and led to the development of warfarin. Other derivatives are being explored for their potential in treating diseases such as cancer due to their ability to interfere with cell proliferation and induce apoptosis in cancer cells. Coumarin also plays a role in agricultural chemistry. It is used in the synthesis of rodenticides that use its anticoagulant properties to control rodent populations. In addition, coumarin derivatives are used as plant growth regulators and antifungal agents, helping to protect crops and increase yields. In materials science, coumarin derivatives are used to develop fluorescent dyes and probes. These compounds exhibit strong fluorescence, making them useful for bioimaging and as markers for various analytical techniques. The versatility of the coumarin chemical structure allows for its modification to tune the fluorescence properties, enhancing its usefulness in scientific research. The presence of coumarin in nature and its wide range of applications highlight its importance in natural and synthetic chemistry. Despite its beneficial uses, it is worth noting that high doses of coumarin may produce hepatotoxicity, which has led regulatory authorities to restrict its use in food and cosmetics. The European Union and the United States have established maximum permissible levels of coumarin in consumer products to ensure safety. References 2010. Unusual recyclization of 2-methyl-1-phenyliso- quinolinium iodide to 1-phenyl-1,4-dihydro- 3h-isochroman-3-one. Chemistry of Heterocyclic Compounds, 46(3). DOI: 10.1007/s10593-010-0516-z 2019. Platinum-on-Carbon-Catalyzed Aqueous Oxidative Lactonization of Diols by Using Molecular Oxygen. Synlett, 30(15). DOI: 10.1055/s-0037-1611917 |
| Market Analysis Reports |