Online Database of Chemicals from Around the World
| Allbiosyn Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allbiosyn.com | |||
 | +86 15900695956 | |||
 | info@allbiosyn.com | |||
 | WeChat: 15900695956 | |||
 | WhatsApp:86 15900695956 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | API >> Inhibitor drug |
|---|---|
| Name | N2-iso-Butyryl-2'-O-(2-methoxyethyl)guanosine |
| Synonyms | N-(9-((2R,3R,4R,5R)-4-Hydroxy-5-(hydroxymethyl)-3-(2-methoxyethoxy)tetrahydrofuran-2-yl)-6-oxo-6,9-dihydro-1H-purin-2-yl)isobutyramide |
| Molecular Structure | 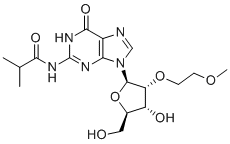 |
| Molecular Formula | C17H25N5O7 |
| Molecular Weight | 411.41 |
| CAS Registry Number | 440327-50-4 |
| SMILES | CC(C)C(=O)NC1=NC2=C(C(=O)N1)N=CN2[C@H]3[C@@H]([C@@H]([C@H](O3)CO)O)OCCOC |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319 Details |
| Safety Statements | P501-P270-P264-P280-P302+P352-P337+P313-P305+P351+P338-P362+P364-P332+P313-P301+P312+P330 Details |
| SDS | Available |
|
N2-isobutyryl-2'-O-(2-methoxyethyl)guanosine (N2-iBu-2'-O-ME-G) is an important compound in nucleoside chemistry that stands out for its structural modifications that enhance its stability and biological activity. This guanosine analog has an isobutyl group at the N2 position and a 2-methoxyethyl group at the 2'-O position. The synthesis of N2-iBu-2'-O-ME-G is part of the development of nucleoside analogs with improved properties for use in biological research. The synthesis involves the introduction of an isobutyl group at the N2 position of guanosine and a 2-methoxyethyl group at the 2'-O position. These modifications increase the resistance of the compound to enzymatic degradation and enhance its solubility in aqueous solutions. The discovery of N2-iBu-2'-O-ME-G stems from the need for more stable and versatile guanosine analogs. Researchers seek to create nucleosides that can better withstand biological processes while maintaining effective interactions with cellular targets. N2-iBu-2'-O-ME-G has several important applications: The isobutyl modification enhances the stability of the guanosine analog, making it useful for studying nucleic acid structure and function. Its enhanced resistance to degradation makes experiments involving RNA and DNA more reliable. N2-iBu-2'-O-ME-G is a valuable tool in drug development, particularly for targeting guanosine-related pathways. Its enhanced stability and modified structure could facilitate the development of novel therapeutics, particularly for diseases involving nucleoside metabolism. The 2-methoxyethyl group improves the cellular uptake of the analog, facilitating studies of nucleoside transport and intracellular distribution. This helps researchers understand how nucleoside analogs are taken up and utilized by cells, which is critical for designing effective drugs. While N2-iBu-2'-O-ME-G is primarily used in research, its properties suggest that it has potential therapeutic applications. Its stability and ability to mimic natural guanosine could be used to develop treatments for diseases involving altered nucleoside metabolism or nucleic acid function, such as viral infections and cancer. References none |
| Market Analysis Reports |