Online Database of Chemicals from Around the World
| Suzhou Biosyntech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biosyntech-suzhou.com | |||
 | +86 13921151340 +86 (512) 6300-1269 | |||
 | sales2@biosyntech-suzhou.com sales@biosyntech-suzhou.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Hubei Innerse Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.innersebio.com | |||
 | +86 (027) 8818-9299 | |||
 | sales@innersebio.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> API intermediate |
|---|---|
| Name | Diethyl (4-isopropyl-3,5-dimethoxybenzyl)phosphonate |
| Synonyms | 5-(diethoxyphosphorylmethyl)-1,3-dimethoxy-2-propan-2-ylbenzene |
| Molecular Structure | 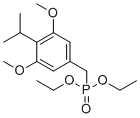 |
| Molecular Formula | C16H27O5P |
| Molecular Weight | 330.36 |
| CAS Registry Number | 443982-76-1 |
| SMILES | CCOP(=O)(CC1=CC(=C(C(=C1)OC)C(C)C)OC)OCC |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.483, Calc.* |
| Boiling Point | 441.7±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 234.3±49.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
Diethyl (4-isopropyl-3,5-dimethoxybenzyl) phosphonate is a well-known organophosphorus compound. Structurally, this compound features benzyl groups substituted with isopropyl and methoxy groups attached to a diethylphosphonate moiety. Its synthesis involves the reaction of benzyl halides with triethyl phosphite followed by functional group modification to achieve the desired isopropyl and methoxy substitutions. The resulting phosphonates are valuable to synthetic chemists due to their stability and reactivity. In organic synthesis, diethyl (4-isopropyl-3,5-dimethoxybenzyl) phosphonate is primarily used in the Horner-Wadsworth-Emmons (HWE) reaction. This reaction forms a carbon-carbon double bond by reacting a phosphonate with an aldehyde or ketone. The HWE reaction is highly regarded for its ability to selectively produce E-olefins, which are essential for the synthesis of complex organic molecules including pharmaceuticals and natural products. The unique substitution pattern on the benzyl ring of diethyl (4-isopropyl-3,5-dimethoxybenzyl) phosphonate imparts specific electronic and steric properties that can influence the reaction processes in which it participates. This makes it a valuable tool for chemists to design pathways to create molecules with precise configurations and functions. The use of this compound extends to serving as an intermediate in the synthesis of bioactive molecules. It can be used to prepare pharmaceuticals, agrochemicals, and fine chemicals, demonstrating the broad applicability of phosphonates in the modern chemical industry. As research in organophosphorus chemistry continues to advance, compounds like diethyl (4-isopropyl-3,5-dimethoxybenzyl) phosphonate remain indispensable for the development of efficient synthetic methods. Their role in promoting complex molecular structures highlights their importance in advancing academic research and industrial applications. References none |
| Market Analysis Reports |