Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hengshui Haoye Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemcoms.com | |||
 | +86 (318) 210-2300 +86 18632882519 | |||
 | +86 (318) 210-3390 | |||
 | firmcn@gmail.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Qingdao Xinnuo Pharmaceutical Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinnuopharma.com | |||
 | +86 (531) 6656-1050 +86 18615688656 +86 18653156686 | |||
 | +86 (531) 6656-1050 | |||
 | sales@xinnuopharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hebei Brant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.brantpharm.com | |||
 | +86 18713118368 | |||
 | lisa@brantpharm.com | |||
 | WhatsApp:0086 18713118368 | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Acid halide |
|---|---|
| Name | 3-(2,6-Dichlorophenyl)-5-methylisoxazole-4-carbonyl chloride |
| Synonyms | 3-(2,6-Dichlorophenyl)-5-methyl-1,2-oxazole-4-carbonyl chloride |
| Molecular Structure | 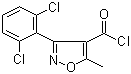 |
| Molecular Formula | C11H6Cl3NO2 |
| Molecular Weight | 290.53 |
| CAS Registry Number | 4462-55-9 |
| EC Number | 224-723-8 |
| SMILES | CC1=C(C(=NO1)C2=C(C=CC=C2Cl)Cl)C(=O)Cl |
| Density | 1.5$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 92 $degree$C (Expl.) |
| Boiling point | 390.9$+/-$42.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 190.2$+/-$27.9 $degree$C (Calc.)* |
| Index of refraction | 1.585 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H315-H319 Details | ||||||||||||||||||||
| Safety Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P351+P338-P305+P354+P338-P316-P321-P332+P317-P337+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-(2,6-Dichlorophenyl)-5-methylisoxazole-4-carbonyl chloride is a functionalized heterocyclic compound that combines an isoxazole ring with a 2,6-dichlorophenyl substituent and a reactive acyl chloride functionality at the 4-position of the isoxazole. Isoxazole derivatives have been widely studied in organic and medicinal chemistry due to their aromatic heterocyclic structure, electron-rich nitrogen-oxygen system, and ability to act as hydrogen-bond acceptors and donors. The acyl chloride group provides a highly reactive site for nucleophilic substitution, making this compound a versatile intermediate for the synthesis of amides, esters, and other functional derivatives. Structurally, the molecule consists of a five-membered isoxazole ring bearing a methyl group at the 5-position, which influences steric and electronic properties. The 3-position is substituted with a 2,6-dichlorophenyl group, adding hydrophobicity, electron-withdrawing character, and potential for π–π interactions. The carbonyl chloride at the 4-position serves as a reactive electrophilic center that can readily form covalent bonds with nucleophiles such as amines, alcohols, or thiols, enabling the preparation of a wide range of functionalized compounds for chemical, material, or pharmaceutical applications. The synthesis of 3-(2,6-dichlorophenyl)-5-methylisoxazole-4-carbonyl chloride generally begins with the corresponding 3-(2,6-dichlorophenyl)-5-methylisoxazole-4-carboxylic acid. Conversion to the acyl chloride is achieved using reagents such as thionyl chloride, oxalyl chloride, or phosphorus pentachloride under controlled conditions. This transformation selectively activates the carboxylic acid while maintaining the integrity of the isoxazole and dichlorophenyl substituents, yielding a stable yet highly reactive intermediate suitable for subsequent reactions. In medicinal chemistry, derivatives of isoxazole-4-carbonyl chlorides are valuable for constructing bioactive amides, which can serve as enzyme inhibitors, receptor ligands, or antimicrobial agents. The dichlorophenyl group enhances hydrophobic interactions with target proteins, while the isoxazole ring provides planarity and hydrogen-bonding capability. The acyl chloride functionality allows for rapid derivatization to produce libraries of compounds for structure–activity relationship studies, facilitating the optimization of potency, selectivity, and pharmacokinetic properties. Beyond pharmaceutical applications, 3-(2,6-dichlorophenyl)-5-methylisoxazole-4-carbonyl chloride is used in organic synthesis as an intermediate for the preparation of heterocyclic compounds, peptidomimetics, and functionalized materials. The carbonyl chloride reacts readily with nucleophiles to form amides, esters, or thioesters, providing access to derivatives with varied electronic and steric properties. The 2,6-dichlorophenyl substituent can also undergo further functionalization through electrophilic aromatic substitution or cross-coupling reactions, expanding the chemical diversity obtainable from this compound. The combination of the isoxazole ring, methyl substitution, dichlorophenyl group, and reactive carbonyl chloride makes this compound both chemically versatile and pharmacologically relevant. Its structural features enable selective nucleophilic reactions, π–π interactions, and hydrogen bonding, which are critical for the design of bioactive molecules, synthetic intermediates, and advanced functional materials. Overall, 3-(2,6-dichlorophenyl)-5-methylisoxazole-4-carbonyl chloride is a multifunctional heterocyclic intermediate that combines an isoxazole scaffold, a hydrophobic dichlorophenyl substituent, and a reactive acyl chloride. Its chemical reactivity and structural properties make it valuable for medicinal chemistry, organic synthesis, and the preparation of bioactive derivatives and functionalized materials. References 2022. A quantitative high-throughput screen identifies compounds that lower expression of the SCA2-and ALS-associated gene ATXN2. The Journal of Biological Chemistry. DOI: 10.1016/j.jbc.2022.102228 2021. A target-agnostic screen identifies approved drugs to stabilize the endoplasmic reticulum-resident proteome. Cell Reports. DOI: 10.1016/j.celrep.2021.109040 |
| Market Analysis Reports |