Online Database of Chemicals from Around the World
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Cheng Du Chenlv Herb Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chenlvherbs.com | |||
 | +86 18981728582 | |||
 | chenlvherb@126.com | |||
 | WeChat: Cheng Du Chenlv Herb Co., Ltd. | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Biochemical >> Inhibitor >> Cytoskeletal signaling >> HSP e.g. HSP90 inhibitor |
|---|---|
| Name | Dihydroumbellatine |
| Synonyms | 5,8-Dihydro-9,10-dimethoxy-6H-benzo[g]-1,3-benzodioxolo[5,6-a]quinolizine |
| Molecular Structure | 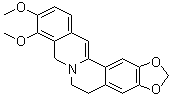 |
| Molecular Formula | C20H19NO4 |
| Molecular Weight | 337.37 |
| CAS Registry Number | 483-15-8 |
| EC Number | 683-111-2 |
| SMILES | COC1=C(C2=C(C=C1)C=C3C4=CC5=C(C=C4CCN3C2)OCO5)OC |
| Solubility | Insoluble (2.2E-3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.37±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 223-224 °C (dec.)** |
| Boiling point | 557.8±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 170.7±27.3 °C (Calc.)* |
| Index of refraction | 1.679 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| ** | Freund, Martin |
| Hazard Symbols |  GHS06 Danger Details GHS06 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300 Details | ||||||||
| Safety Statements | P264-P270-P301+P316-P321-P330-P405-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Dihydroumbellatine is a naturally occurring coumarin derivative that belongs to the broader family of phenylpropanoid compounds. It is primarily isolated from certain plants in the Umbelliferae (Apiaceae) family and other botanical sources known for their rich coumarin content. Structurally, dihydroumbellatine is characterized by a lactone ring fused to an aromatic system, with additional hydrogenation compared to its parent compound umbellatine, resulting in reduced double bonds and altered electronic properties that can influence its biological activity. The discovery of dihydroumbellatine originated from phytochemical investigations aimed at identifying bioactive constituents in medicinal plants used traditionally for gastrointestinal, anti-inflammatory, and antimicrobial purposes. Researchers isolated and characterized the compound using chromatographic separation techniques and spectroscopic methods, which facilitated structural elucidation and enabled further studies of its pharmacological effects. Pharmacologically, dihydroumbellatine exhibits a range of biological activities. It has demonstrated anti-inflammatory effects by modulating the production of pro-inflammatory mediators and influencing cellular signaling pathways involved in inflammation. This property has made it a subject of interest for potential applications in managing inflammatory disorders and mitigating tissue damage caused by oxidative stress and inflammation. In addition to its anti-inflammatory activity, dihydroumbellatine has been studied for its antioxidant properties. It is capable of scavenging reactive oxygen species and protecting cellular components from oxidative damage, which may contribute to its protective effects in various tissues. Such antioxidant activity supports its potential utility in reducing oxidative stress–related pathologies and promoting overall cellular health. Dihydroumbellatine has also shown preliminary antimicrobial activity in in vitro studies. It exhibits inhibitory effects against certain bacterial and fungal strains, suggesting potential applications as a natural antimicrobial agent or as a lead compound for the development of new antimicrobial drugs. These properties, combined with its relatively low toxicity in preliminary studies, make it a promising candidate for further pharmacological exploration. The bioavailability and pharmacokinetic profile of dihydroumbellatine are still under investigation, as with many naturally occurring coumarins. Research into formulation strategies, such as enhancing solubility or stability, may improve its therapeutic potential and enable effective delivery in medicinal or nutraceutical applications. Clinically, while dihydroumbellatine has not yet reached widespread therapeutic use, its diverse biological activities indicate potential applications in inflammation, oxidative stress, and microbial infections. Ongoing research focuses on elucidating its molecular mechanisms, optimizing its pharmacological properties, and evaluating safety and efficacy in preclinical models. Overall, dihydroumbellatine is a bioactive coumarin derivative with anti-inflammatory, antioxidant, and antimicrobial properties. Its structural characteristics, combined with traditional use of its plant sources and emerging pharmacological data, highlight its potential as a compound of interest in both medicinal and nutraceutical contexts. Continued research is essential to fully establish its therapeutic applications and to develop strategies for effective delivery and clinical use. References 2019. Comparison of anti-inflammatory effects of berberine, and its natural oxidative and reduced derivatives from Rhizoma Coptidis in vitro and in vivo. Phytomedicine : international journal of phytotherapy and phytopharmacology, 52. DOI: 10.1016/j.phymed.2018.09.228 2019. Dihydroberberine, a hydrogenated derivative of berberine firstly identified in Phellodendri Chinese Cortex, exerts anti-inflammatory effect via dual modulation of NF-κB and MAPK signaling pathways. International Immunopharmacology, 75. DOI: 10.1016/j.intimp.2019.105802 2021. Dihydroberberine, an isoquinoline alkaloid, exhibits protective effect against dextran sulfate sodium-induced ulcerative colitis in mice. Phytomedicine : international journal of phytotherapy and phytopharmacology, 90. DOI: 10.1016/j.phymed.2021.153631 |
| Market Analysis Reports |