Online Database of Chemicals from Around the World
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shenzhen Geneseqtools Bioscience & Technology Co. Ltd. | China | |||
|---|---|---|---|---|
 | geneseqtools.com | |||
 | +86 (0755) 2325-1735 | |||
 | sales@geneseqtools.com | |||
 | QQ Chat | |||
 | WeChat: 13302967066 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| SBS Genetech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sbsbio.com | |||
 | +86 (10) 8278-4296 8278-4292 6296-9345 | |||
 | +86 (10) 8278-4290 | |||
 | info@sbsbio.com | |||
| Chemical manufacturer | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Classification | Biochemical >> Carbohydrate >> Monosaccharide |
|---|---|
| Name | 3-Indoxyl-beta-D-glucopyranoside |
| Synonyms | 3-Indoxyl-beta-D-glucoside |
| Molecular Structure | 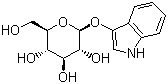 |
| Molecular Formula | C14H17NO6 |
| Molecular Weight | 295.29 |
| CAS Registry Number | 487-60-5 |
| EC Number | 610-433-2 |
| SMILES | C1=CC=C2C(=C1)C(=CN2)O[C@H]3[C@@H]([C@H]([C@@H]([C@H](O3)CO)O)O)O |
| Density | 1.6±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 57 - 58 °C (Expl.) |
| Boiling point | 606.7±55.0 °C 760 mmHg (Calc.)* |
| Flash point | 320.7±31.5 °C (Calc.)* |
| Solubility | H2O: 0.1 M (20 °C) (Expl.) |
| Index of refraction | 1.715 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details |
| Safety Statements | P261-P280-P305+P351+P338 Details |
| SDS | Available |
|
3-Indoxyl-β-D-glucopyranoside is a synthetic chromogenic substrate widely used in biochemical assays to detect the activity of β-D-glucosidase enzymes. The molecule consists of an indoxyl moiety linked through an oxygen bridge to a β-D-glucopyranoside group. This glycosidic linkage is specifically cleaved by β-D-glucosidase, releasing the indoxyl group, which can then undergo oxidative dimerization to form an intensely colored compound. The resulting color change allows for easy visual or spectrophotometric detection of enzymatic activity. The discovery and development of 3-indoxyl-β-D-glucopyranoside are rooted in the search for sensitive and selective substrates for glycosidase enzymes. Glycosidases, including β-glucosidase, play a vital role in various physiological and pathological processes, such as carbohydrate metabolism, lysosomal function, and microbial fermentation. The need to study these enzymes in both diagnostic and research contexts led to the creation of synthetic glycosides that release easily detectable products upon enzymatic hydrolysis. Upon enzymatic cleavage by β-D-glucosidase, 3-indoxyl-β-D-glucopyranoside yields 3-hydroxyindole (commonly referred to as indoxyl). In the presence of atmospheric oxygen or mild oxidizing agents, indoxyl undergoes spontaneous oxidation to form indigo blue, a highly colored and insoluble dye. This colorimetric change serves as the basis for many applications involving the detection of β-glucosidase activity. In microbiology, for example, the compound is commonly incorporated into growth media to differentiate bacterial species based on their ability to produce β-glucosidase. Colonies of organisms expressing the enzyme appear blue or blue-green due to indigo formation, allowing rapid visual identification. In clinical diagnostics, 3-indoxyl-β-D-glucopyranoside is used in assays to detect β-glucosidase deficiencies or to monitor the activity of lysosomal enzymes in certain metabolic disorders. One such example is Gaucher's disease, a lysosomal storage disorder caused by a deficiency of β-glucosidase. The substrate can be used in biochemical assays to assess enzyme function in blood or tissue samples, supporting diagnosis and treatment monitoring. The compound is also employed in food and environmental testing. In the food industry, it serves as a tool to detect microbial contamination by identifying β-glucosidase-positive bacteria. In environmental microbiology, it is used to monitor microbial populations in soil or water samples, particularly those involved in organic matter degradation and nutrient cycling. An advantage of using 3-indoxyl-β-D-glucopyranoside in detection systems is the intensity and stability of the resulting indigo color. This allows for reliable and reproducible results in both qualitative and quantitative assays. The substrate is generally stable under standard laboratory storage conditions and exhibits good solubility in aqueous media, making it practical for routine use. Furthermore, the use of this substrate in chromogenic media has enhanced the development of selective and differential culture media. By combining 3-indoxyl-β-D-glucopyranoside with other enzyme substrates or growth indicators, researchers and clinicians can simultaneously test for multiple microbial traits, increasing the diagnostic power of simple plate-based methods. Overall, 3-indoxyl-β-D-glucopyranoside has proven to be a highly valuable reagent for the specific detection of β-D-glucosidase activity. Its utility in microbiology, enzymology, clinical diagnostics, and environmental testing underscores its importance in both research and applied science. The clarity and intensity of the colorimetric response, combined with the specificity of the enzymatic reaction, make it a preferred choice in many standard biochemical assays. References 1988. Indoxyl-β-D-glucuronide, a novel chromogenic reagent for the specific detection and enumeration of Escherichia coli in environmental samples. Canadian Journal of Microbiology, 34(5). DOI: 10.1139/m88-115 1990. Biosynthesis of indoxyl derivatives in Isatis tinctoria and Polygonum tinctorium. Phytochemistry, 29(10). DOI: 10.1016/0031-9422(90)80025-c 2001. Identification of an indigo precursor from leaves of Isatis tinctoria (Woad). Phytochemistry, 58(6). DOI: 10.1016/s0031-9422(01)00335-1 |
| Market Analysis Reports |