Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Alway Chem, China | China | |||
|---|---|---|---|---|
 | www.alwaychem.net | |||
 | +86 (532) 8586-4000 | |||
 | +86 (532) 8589-3133 | |||
 | alwaychem@gmail.com alwaychem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zhejiang Dongdong Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dongdong-chem.com | |||
 | +86 (576) 8569-8599 | |||
 | melon522@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Taicang Qianjing Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qjchem.com | |||
 | +86 (512) 5364-5646 | |||
 | +86 (512) 5364-5227 | |||
 | sales@qjchem.com tcqxh@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Kendu Pharmchem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kenduchem.com | |||
 | +86 (571) 8605-9031 | |||
 | +86 (571) 8605-9210 | |||
 | kdpharmchem@gmail.com | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 5-Chloro-1-methyl-4-nitroimidazole |
| Synonyms | 5-Chloro-1-methyl-4-nitro-1H-imidazole |
| Molecular Structure | 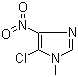 |
| Molecular Formula | C4H4ClN3O2 |
| Molecular Weight | 161.55 |
| CAS Registry Number | 4897-25-0 |
| EC Number | 225-521-2 |
| SMILES | CN1C=NC(=C1Cl)[N+](=O)[O-] |
| Density | 1.7±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 148-150 °C (Expl.) |
| Index of Refraction | 1.648, Calc.* |
| Boiling Point | 362.3±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 172.9±22.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
5-Chloro-1-methyl-4-nitroimidazole is an organic compound that belongs to the class of imidazole derivatives. It has the molecular formula C5H5ClN2O2 and consists of a chlorinated imidazole ring with a methyl group at position 1 and a nitro group at position 4. This compound has been studied for its chemical properties and its potential applications in various fields. The synthesis of 5-chloro-1-methyl-4-nitroimidazole was first reported in the literature in the mid-20th century as part of research on substituted imidazoles. The imidazole ring structure, with its nitrogen atoms, allows for a wide range of reactivity, making such compounds useful intermediates in organic synthesis. The specific substitutions at positions 1, 4, and 5 of the imidazole ring introduce unique electronic and steric properties that impact the reactivity of the molecule. 5-Chloro-1-methyl-4-nitroimidazole has been studied for its potential as an intermediate in the synthesis of pharmaceutical and agricultural compounds. One area of application is in the synthesis of antifungal and antimicrobial agents. The imidazole ring structure is known for its biological activity, particularly as a component of molecules that exhibit antifungal, antibacterial, or antiprotozoal properties. The presence of the chloro and nitro groups in 5-chloro-1-methyl-4-nitroimidazole may influence its biological activity, though the specific interactions and mechanisms of action have been explored in various studies. In addition to its role in pharmaceutical synthesis, this compound has potential applications in agrochemicals, particularly in the development of fungicides and herbicides. The imidazole structure is a key feature of several fungicidal compounds, and the introduction of specific substituents such as chloro and nitro groups can enhance the compound's effectiveness or selectivity. Studies have indicated that imidazole derivatives can disrupt the cell membranes of fungi or inhibit essential enzymes involved in their growth and replication. Another important area where 5-chloro-1-methyl-4-nitroimidazole has been explored is in the field of organic electronics. Imidazole derivatives are sometimes incorporated into organic semiconductor materials due to their ability to participate in electron transfer processes. The substitution of functional groups like chloro and nitro may affect the electronic properties of these compounds, making them useful in organic electronics such as organic light-emitting diodes (OLEDs) or organic solar cells (OPVs). These materials are being actively researched for their potential in energy-efficient devices. 5-Chloro-1-methyl-4-nitroimidazole has also been studied in the context of synthetic chemistry as a reagent or catalyst. Its specific reactivity due to the electron-withdrawing nitro group and the electron-donating methyl group makes it a potential candidate for various reactions, including those that involve nucleophilic substitution or other types of functionalization of organic molecules. In conclusion, 5-chloro-1-methyl-4-nitroimidazole is a compound with applications in pharmaceuticals, agrochemicals, organic electronics, and synthetic chemistry. Its properties as an imidazole derivative, along with the presence of chloro and nitro groups, make it valuable for the synthesis of biologically active molecules, electronic materials, and other chemical intermediates. References 1980. Rapid-mix Studies on the Anomalous Radiosensitization of Mammalian Cells by 5-chloro-1-methyl-4-nitromidazole. International Journal of Radiation Biology and Related Studies in Physics, Chemistry, and Medicine, 37(1). DOI: 10.1080/09553008014551501 2009. New Synthesis and Antiparasitic Activity of Model 5-Aryl-1-methyl-4-nitroimidazoles. Molecules, 14(8). DOI: 10.3390/molecules14082758 2016. 4-Cyclopropyl-1-(1-methyl-4-nitro-1H-imidazol-5-yl)-1H-1,2,3-triazole and Ethyl 1-(1-methyl-4-nitro-1H-imidazol-5-yl)-1H-1,2,3-triazole-4-carboxylate. Journal of Chemical Crystallography, 46(7). DOI: 10.1007/s10870-016-0659-6 |
| Market Analysis Reports |