Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Shanghai Nanxiang Reagent Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nx-reagent.com | |||
 | +86 (21) 3912-7448 +86 13512139665 | |||
 | +86 (21) 3912-7431 | |||
 | xumiao@nx-reagent.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2007 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Suqian Nanxiang Chemicals Manufacturing Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nanxiangchem.com | |||
 | +86 (0527) 8483-6748 | |||
 | sales@nanxiangchem.com | |||
 | QQ Chat | |||
 | WeChat: 13512139665 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | 3-Bromopropylamine hydrobromide |
| Synonyms | 3-Aminopropyl bromide hydrobromide |
| Molecular Structure | 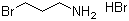 |
| Molecular Formula | C3H8BrN.HBr |
| Molecular Weight | 218.92 |
| CAS Registry Number | 5003-71-4 |
| EC Number | 225-675-0 |
| SMILES | C(CN)CBr.Br |
| Melting point | 171-175 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-Bromopropylamine hydrobromide is an organic compound with the molecular formula C3H9Br2N, characterized by the presence of both a bromine atom and an amine group attached to a three-carbon chain. The discovery of brominated alkylamines like 3-bromopropylamine hydrobromide has its roots in early organohalide chemistry, where halogenation of amines was explored to alter their reactivity and introduce new functionalities. Bromine atoms are commonly introduced into organic molecules through substitution reactions, providing a versatile handle for further chemical transformations. 3-Bromopropylamine hydrobromide is synthesized through the bromination of propylamine or via the reaction of propylamine with hydrogen bromide and bromine. The hydrobromide salt form enhances the stability and handling of the compound, making it more convenient for storage and use in laboratory settings. The bromine atom in the 3-position of the alkyl chain offers a site for nucleophilic substitution, while the primary amine provides a reactive group for coupling with other molecules, making this compound a useful intermediate in organic synthesis. The applications of 3-bromopropylamine hydrobromide span various fields, particularly in pharmaceuticals, biochemistry, and materials science. In pharmaceutical research, it is frequently employed as a key intermediate for the synthesis of biologically active molecules. The bromine atom allows for subsequent functionalization through nucleophilic substitution reactions, enabling the creation of new carbon-nitrogen bonds, which are central to the synthesis of many drug molecules. In medicinal chemistry, alkylamines are important building blocks in the development of pharmaceutical agents, and the brominated form allows for the selective introduction of amine groups into target molecules. This makes 3-bromopropylamine hydrobromide a useful reagent in the preparation of drugs and drug precursors. In biochemistry, 3-bromopropylamine hydrobromide can be used to modify biomolecules such as peptides and proteins through alkylation reactions. The amine group can react with carboxylic acids or other nucleophilic groups in biomolecules, while the bromine can be displaced by nucleophiles, allowing for the attachment of the alkyl chain to various biological targets. This has potential applications in studying enzyme mechanisms, protein labeling, and developing new bioactive compounds. Furthermore, in the field of materials science, 3-bromopropylamine hydrobromide serves as a building block for the synthesis of polymers, resins, and other advanced materials. The ability to introduce alkylamine functionalities into polymers provides avenues for creating materials with tailored properties, such as enhanced mechanical strength or specific reactivity. It is particularly useful in the creation of functionalized surfaces and coatings, where the amine group can participate in further reactions to modify the surface properties of materials. Overall, 3-bromopropylamine hydrobromide is a valuable chemical intermediate with a wide range of applications in organic synthesis, pharmaceutical development, biochemistry, and materials science. Its discovery has provided chemists with a versatile tool for introducing both halogen and amine functionalities into various molecular frameworks, facilitating the creation of complex, functional molecules. References 2019. Immobilized amino-containing basic ionic liquid on graphene oxide as an efficient and stable catalyst for transesterification. Reaction Kinetics, Mechanisms and Catalysis. DOI: 10.1007/s11144-019-01589-7 2016. Salts of 2- or 3-haloalkylamines in the synthesis of N-aminoalkyl derivatives of heterocyclic and aromatic amines. Russian Chemical Bulletin. DOI: 10.1007/s11172-016-1570-1 |
| Market Analysis Reports |