Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Pharmaceutical intermediate >> OLED material intermediate |
|---|---|
| Name | 3-Iodo-9-phenylcarbazole |
| Synonyms | 3-Iodo-N-phenylcarbazole |
| Molecular Structure | 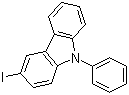 |
| Molecular Formula | C18H12IN |
| Molecular Weight | 369.20 |
| CAS Registry Number | 502161-03-7 |
| EC Number | 808-151-1 |
| SMILES | C1=CC=C(C=C1)N2C3=C(C=C(C=C3)I)C4=CC=CC=C42 |
| Solubility | 0.001784 mg/L (25 °C water) |
|---|---|
| Density | 1.6±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.704, Calc.* |
| Melting point | 171.22 °C |
| Boiling Point | 480.6±27.0 °C (760 mmHg), Calc.*, 434.60 °C |
| Flash Point | 244.5±23.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
3-Iodo-9-phenylcarbazole is an organic compound that belongs to the family of carbazole derivatives. It is characterized by the presence of an iodine atom at the 3-position of the carbazole ring and a phenyl group attached to the nitrogen of the carbazole moiety. This structure imparts unique chemical and electronic properties, making it a valuable compound in various fields, including organic electronics and materials science. The discovery of 3-iodo-9-phenylcarbazole is part of the broader exploration of carbazole derivatives, which have attracted significant attention due to their high stability, strong aromaticity, and versatile reactivity. Carbazoles themselves have been known for over a century, with early studies focused on their aromaticity and use as building blocks in organic synthesis. The introduction of iodine at the 3-position of the carbazole ring enhances the compound's electronic properties and makes it more reactive in certain reactions, opening up new avenues for its use in both fundamental and applied chemistry. Synthesis of 3-iodo-9-phenylcarbazole is typically achieved through halogenation reactions involving carbazole derivatives. The iodine atom is often introduced via electrophilic aromatic substitution, where iodine is used as a halogenating agent. The presence of the phenyl group at the nitrogen position also contributes to the compound’s stability and enhances its solubility in organic solvents, which is beneficial for various chemical reactions and applications. One of the most important applications of 3-iodo-9-phenylcarbazole is in the field of organic electronics. It serves as a key intermediate in the development of organic semiconductors, specifically in the fabrication of organic light-emitting diodes (OLEDs) and organic solar cells. The iodine atom in the structure plays a crucial role in modifying the electronic properties of the molecule, improving charge transport and enhancing the efficiency of these devices. Additionally, carbazole derivatives like 3-iodo-9-phenylcarbazole are often used in the development of hole transport materials (HTMs) in OLEDs and organic photovoltaics (OPVs), where they help in the effective movement of charge carriers, thus contributing to the performance of these devices. In addition to its role in electronic applications, 3-iodo-9-phenylcarbazole has potential applications in medicinal chemistry. The carbazole core is known to exhibit a wide range of biological activities, including antimicrobial, anticancer, and anti-inflammatory effects. Research into the pharmacological properties of carbazole derivatives has led to the exploration of compounds like 3-iodo-9-phenylcarbazole for use in drug development. The iodine atom, in particular, can enhance the lipophilicity of the compound, potentially improving its ability to cross biological membranes and interact with target sites. References 2022. Recent Progress on Transition-Metal-Mediated Reductive C(sp3)-O Bond Radical Addition and Coupling Reactions. Synthesis, 54(13). DOI: 10.1055/a-1848-3005 |
| Market Analysis Reports |