Online Database of Chemicals from Around the World
| Zouping Mingxing Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zoutong.com.cn | |||
 | +86 13605431940 | |||
 | +86 (543) 224-0079 | |||
 | sdzpmxchem@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Wuxi Orient Detergent Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinobromide.com | |||
 | +86 (510) 8758-1290 | |||
 | +86 (510) 8758-3694 | |||
 | suguangming@126.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Haimen Best Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bestfine.com | |||
 | +86 (513) 8538-3385 +86 13506281858 | |||
 | +86 (513) 8538-3386 | |||
 | best@bestfine.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| La Mesta Chimie Fine S.A.S. | France | |||
|---|---|---|---|---|
 | www.la-mesta.com | |||
 | +33 (4) 9208-5300 | |||
 | +33 (4) 9208-5340 | |||
 | info@la-mesta.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Reddy N Reddy Pharmaceuticals | India | |||
|---|---|---|---|---|
 | www.reddynreddypharma.com | |||
 | +91 (40) 2307-5686 +91 98480-96759 +91 98481-96759 | |||
 | info@reddynreddypharma.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Henan Yuchen Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuchenpharm.com | |||
 | +86 (374) 569-9606 | |||
 | +86 (374) 569-9958 | |||
 | yc999@china.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2021 | ||||
| Zoupingtongfengchemical Co. Ltd. | China | |||
|---|---|---|---|---|
 | www.tongfeng-chem.com | |||
 | +86 18769685991 | |||
 | tongfengchem@163.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Mendel Chemicals | Moldova | |||
|---|---|---|---|---|
 | www.mendelchemicals.com | |||
 | +373 78813683 | |||
 | info@mendelchemicals.com | |||
 | WhatsApp:+37378813683 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Anhui Weixiang New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ahwxchem.com | |||
 | +86 13195623283 | |||
 | export@ahwxchem.com | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Ningbo Yide Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yide-chem.com | |||
 | +86 (574) 6258-9038 6258-9338 | |||
 | +86 (574) 6258-7520 | |||
 | yd@yide-chem.com | |||
| Chemical manufacturer | ||||
| ChemSampCo, Inc. | USA | |||
|---|---|---|---|---|
 | www.chemsampco.com | |||
 | +1 (609) 656-2440 | |||
 | +1 (609) 656-2446 | |||
 | sales@chemsampco.com | |||
| Chemical manufacturer since 1960 | ||||
| Jiangsu Tendenci Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tendencichem.com | |||
 | +86 (512) 5283-8508 | |||
 | +86 (512) 5283-8508 | |||
 | tjp@tendencichem.com | |||
| Chemical manufacturer since 1983 | ||||
| Classification | Chemical reagent >> Organic reagent >> Halogenated aliphatic hydrocarbon |
|---|---|
| Name | 2-Chloro-2-methylpropane |
| Synonyms | 2-Chloro-2-methyl propane; tert-Butyl chloride; Trimethylchloromethane |
| Molecular Structure | 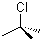 |
| Molecular Formula | C4H9Cl |
| Molecular Weight | 92.57 |
| CAS Registry Number | 507-20-0 |
| EC Number | 208-066-4 |
| SMILES | CC(C)(C)Cl |
| Density | 0.9±0.1 g/cm3 Calc.*, 0.851 g/mL (Expl.) |
|---|---|
| Melting point | -25 °C (Expl.) |
| Boiling point | 50.6 °C 760 mmHg (Calc.)*, 51 - 52 °C (Expl.) |
| Flash point | 18.3 °C (Calc.)*, -23 °C (Expl.) |
| Solubility | water slightly soluble (Expl.) |
| Index of refraction | 1.395 (Calc.)*, 1.385 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS02 Danger Details GHS02 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P280-P303+P361+P353-P370+P378-P403+P235-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 1127 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
2-Chloro-2-methylpropane, also known as tert-butyl chloride, is a branched alkyl halide with the molecular formula C4H9Cl. It appears as a colorless, flammable liquid with a characteristic odor and is recognized for its role as a reactive intermediate in organic synthesis. The compound consists of a chlorine atom bonded to a tertiary carbon, which is connected to three methyl groups, making it highly susceptible to nucleophilic substitution and elimination reactions. The compound was first synthesized in the 19th century through the chlorination of tert-butyl alcohol with hydrogen chloride under controlled conditions. The tertiary carbon structure stabilizes the carbocation intermediate, facilitating SN1-type reactions and making the compound an important model for studying carbocation chemistry and reaction mechanisms in branched systems. Industrial preparation focuses on optimizing yield while minimizing side reactions such as rearrangement or elimination to form isobutylene. Chemically, 2-chloro-2-methylpropane is used extensively as a precursor for tert-butyl derivatives in both laboratory and industrial settings. Its reactivity allows for the formation of ethers, amines, esters, and other functional groups through nucleophilic substitution. The tertiary nature of the carbon center favors SN1 reactions over SN2, providing selective pathways for synthesis. This property is particularly valuable in preparing compounds that require minimal steric hindrance during substitution. One of the main industrial applications of 2-chloro-2-methylpropane is in the production of tert-butyl ethers, such as methyl tert-butyl ether (MTBE), which historically served as a gasoline additive to increase octane rating and improve combustion efficiency. It is also employed in synthesizing antioxidants, stabilizers, and other specialty chemicals where the tert-butyl group imparts steric bulk and influences reactivity, solubility, and stability of the resulting molecules. In pharmaceutical chemistry, 2-chloro-2-methylpropane functions as a key intermediate in the synthesis of various active pharmaceutical ingredients. Its ability to introduce a bulky tert-butyl group into molecules can enhance metabolic stability and modify lipophilicity, contributing to improved pharmacokinetic properties. Similarly, it is used in agrochemical applications to prepare herbicides, pesticides, and other crop protection agents, where the tert-butyl moiety helps tailor molecular behavior and efficacy. Mechanistically, the tertiary chloride provides a valuable system for studying carbocation formation and the influence of steric and electronic effects on reaction kinetics. Its behavior under acidic and nucleophilic conditions offers insight into reaction pathways and the development of selective synthetic strategies. The compound’s volatility and moderate boiling point also facilitate its use in vapor-phase reactions and distillation processes. Physically, 2-chloro-2-methylpropane is insoluble in water but miscible with most organic solvents, including ethers, alcohols, and hydrocarbons. It exhibits a relatively low vapor pressure and is flammable, requiring careful handling in laboratory and industrial environments. Exposure precautions include using gloves, eye protection, and proper ventilation, as the compound can cause irritation and adverse health effects if inhaled or absorbed. Environmental considerations focus on preventing release into water and soil due to its potential toxicity. The discovery and utilization of 2-chloro-2-methylpropane highlight its significance as a reactive tertiary alkyl halide. Its chemical versatility, ability to undergo selective substitution reactions, and functional role in the synthesis of ethers, pharmaceuticals, and specialty chemicals ensure its ongoing importance in organic chemistry and industrial applications. References 2022. A Review on the Halodefluorination of Aliphatic Fluorides. Synthesis, 54(4). DOI: 10.1055/a-1684-0121 2025. Mechanochemical activation of metallic lithium for the generation and application of organolithium compounds in air. Nature Synthesis, 4(3). DOI: 10.1038/s44160-025-00753-3 2023. A highly ionic reaction environment achieving low-temperature polyethylene upcycling. Science China Chemistry, 66(6). DOI: 10.1007/s11426-023-1561-y |
| Market Analysis Reports |