Online Database of Chemicals from Around the World
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Selleck Chemicals LLC | USA | |||
|---|---|---|---|---|
 | www.selleckchem.com | |||
 | +1 (713) 535-9129 | |||
 | +1 (832) 582-8590 | |||
 | info@selleckchem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Targetmol China | China | |||
|---|---|---|---|---|
 | www.targetmol.cn | |||
 | +86 (400) 820-0310 | |||
 | sales@targetmol.cn | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2025 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| VDM Biochemicals | USA | |||
|---|---|---|---|---|
 | www.vdmbio.com | |||
 | +1 (330) 252-8181 | |||
 | +1 (330) 252-8171 | |||
 | sales@vdmbio.com info@vdmbio.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Nucleoside drugs >> Nucleotides and their analogues |
|---|---|
| Name | 6-Amino-3-methylpurine |
| Synonyms | 3-Methyladenine; 3-Methyl-6-aminopurine; N3-Methyladenine; NSC 66389 |
| Molecular Structure | 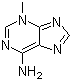 |
| Molecular Formula | C6H7N5 |
| Molecular Weight | 149.15 |
| CAS Registry Number | 5142-23-4 |
| EC Number | 225-908-6 |
| SMILES | CN1C=NC(=N)C2=C1N=CN2 |
| Density | 1.6±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 300 °C (Decomposes) (Expl.) |
| Boiling point | 240.1±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 99.0±30.1 °C (Calc.)* |
| Solubility | Soluble 50 mM in DMSO (Expl.) |
| Index of refraction | 1.807 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319 Details | ||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
6-Amino-3-methylpurine is a purine derivative characterized by the presence of an amino group at the 6-position and a methyl group at the 3-position of the purine ring. Purines are heterocyclic aromatic organic compounds that play essential roles in biological systems, forming the structural basis for important biomolecules such as adenine and guanine, which are components of nucleic acids. The discovery of 6-amino-3-methylpurine stems from research into purine analogs and their biological activity. Modifications of the purine structure have been extensively studied to understand nucleic acid chemistry, enzyme interactions, and as potential therapeutic agents. 6-Amino-3-methylpurine is one such synthetic analog that has been used in biochemical and pharmacological studies. This compound exhibits properties relevant to nucleic acid biochemistry, as it can mimic natural purines in certain contexts or act as an inhibitor of enzymes involved in purine metabolism. Its structural modifications affect its ability to interact with enzymes such as purine nucleoside phosphorylase and other purine-metabolizing enzymes, making it valuable in studying metabolic pathways and enzyme specificity. In medicinal chemistry, purine analogs like 6-amino-3-methylpurine have been investigated for their potential as antimetabolites, antiviral agents, and anticancer drugs. Although 6-amino-3-methylpurine itself is not widely used as a drug, its structural framework serves as a basis for the design of compounds that interfere with DNA and RNA synthesis, thus impacting cell proliferation. The applications of 6-amino-3-methylpurine are primarily experimental and research-oriented. It is utilized as a biochemical tool to probe enzyme function, study nucleotide metabolism, and in the synthesis of modified nucleotides. Its ability to participate in hydrogen bonding and base pairing makes it useful in nucleic acid chemistry for the design of novel oligonucleotides or nucleotide analogs with altered properties. Furthermore, 6-amino-3-methylpurine has been used in studies of mutagenesis and DNA repair mechanisms, as the incorporation of modified purines into DNA can lead to altered base pairing and biological outcomes. This property helps researchers understand the structural and functional consequences of nucleobase modifications in genetic material. Due to its synthetic nature, 6-amino-3-methylpurine is produced through chemical synthesis involving purine ring construction and subsequent substitution reactions. Its purity and stability under laboratory conditions make it suitable for detailed biochemical assays and structural studies. In summary, 6-amino-3-methylpurine is a synthetic purine analog valued for its role in biochemical research, particularly in the study of purine metabolism, enzyme interaction, and nucleic acid chemistry. While it is not directly applied as a therapeutic agent, it contributes significantly to the understanding of purine biology and the development of related pharmaceuticals. References 1994. Purification and Biochemical Characterization of Recombinant N-Methylpurine-DNA Glycosylase of the Mouse. Biochemistry, 33(48). DOI: 10.1021/bi00254a024 2012. Direct Analysis of 5-Methylcytosine and 5-Methyl-2′-deoxycytidine in Human Urine by Isotope Dilution LC-MS/MS: Correlations with N-Methylated Purines and Oxidized DNA Lesions. Chemical Research in Toxicology, 25(2). DOI: 10.1021/tx2004954 2012. Elevated N3-methylpurine-DNA glycosylase DNA repair activity is associated with lung cancer. Mutation Research/Fundamental and Molecular Mechanisms of Mutagenesis, 732(1-2). DOI: 10.1016/j.mrfmmm.2012.01.001 |
| Market Analysis Reports |