In the annals of pharmaceutical history, few compounds have transformed medical practice as profoundly as cimetidine, a histamine H2-receptor antagonist that revolutionized the treatment of acid-related gastrointestinal disorders. Introduced in the 1970s, cimetidine (chemical formula: C10H16N6S) emerged as a pioneering drug, offering a novel approach to managing conditions like peptic ulcers and gastroesophageal reflux disease (GERD). Its discovery and subsequent applications underscore the power of targeted pharmacological innovation and its lasting impact on patient care.
Cimetidine’s journey began at Smith Kline & French (now part of GlaxoSmithKline) in the United Kingdom, where chemist James Black and his team sought to address the limitations of existing treatments for peptic ulcers, such as antacids, which offered only temporary relief. Building on Black’s earlier success with beta-blockers, the team turned their attention to histamine, a key regulator of gastric acid secretion. By the late 1960s, they identified two distinct histamine receptors: H1, linked to allergic responses, and H2, responsible for stimulating acid production in the stomach’s parietal cells. This insight paved the way for the development of H2-receptor antagonists, with cimetidine emerging as the first clinically viable candidate after years of synthesizing and testing hundreds of compounds. Launched in 1976 under the brand name Tagamet, it marked a paradigm shift from symptom management to directly inhibiting acid secretion at its source.
The discovery process was meticulous. Early attempts to block histamine’s action were thwarted by the lack of specificity in antagonists, but Black’s team eventually synthesized cimetidine, a guanidine derivative with a cyanoguanidine group, which selectively targeted H2 receptors without affecting H1 pathways. Its efficacy was demonstrated in clinical trials, showing significant ulcer healing rates and reductions in acid output, a breakthrough detailed in early studies published in The Lancet. This success earned James Black a share of the 1988 Nobel Prize in Physiology or Medicine, recognizing his contributions to rational drug design.
Cimetidine’s applications quickly expanded beyond peptic ulcers. It became a cornerstone therapy for GERD, alleviating symptoms like heartburn by reducing acid reflux into the esophagus. It also found use in managing Zollinger-Ellison syndrome, a rare condition characterized by excessive acid production due to gastrin-secreting tumors. Administered orally or intravenously, cimetidine’s ability to suppress both basal and meal-stimulated acid secretion offered patients a reprieve from debilitating symptoms and reduced the need for invasive surgeries like vagotomy. Its pharmacokinetic profile—rapid absorption and a half-life of about two hours—made it adaptable for both acute and chronic use.
However, cimetidine’s journey was not without challenges. Its inhibition of cytochrome P450 enzymes in the liver, a side effect of its imidazole ring, led to drug interactions with agents like warfarin and theophylline, necessitating careful monitoring. This limitation spurred the development of successors like ranitidine and famotidine, which offered similar efficacy with fewer interactions. Despite this, cimetidine remains a valuable tool in specific contexts, particularly where cost and availability are concerns, and its legacy endures in pharmacology education as a case study in drug discovery.
Today, while proton pump inhibitors (PPIs) have largely supplanted H2 antagonists for acid suppression, cimetidine’s role in medical history is undeniable. Its development illuminated the potential of receptor-targeted therapies, influencing subsequent innovations across therapeutic areas. From its serendipitous origins to its widespread clinical impact, cimetidine exemplifies how scientific curiosity, paired with rigorous research, can yield transformative health solutions.
References
Brimblecombe, R. W., Duncan, W. A. M., Durant, G. J., Emmett, J. C., Ganellin, C. R., and Parsons, M. E., 1975. Cimetidine–a non-thiourea H2-receptor antagonist. Journal of International Medical Research, 3(2), pp. 86-91.
DOI: 10.1177/030006057500300205
Bodemar, G., Norlander, B., and Walan, A., 1979. Cimetidine in the treatment of active peptic ulcer disease. Scandinavian Journal of Gastroenterology, 14(5), pp. 577-584.
Freston, J. W., 1982. Cimetidine: I. Developments, pharmacology, and clinical efficacy. Annals of Internal Medicine, 97(4), pp. 573-580.
|




















































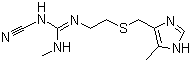

 GHS05;GHS08 Danger Details
GHS05;GHS08 Danger Details