Online Database of Chemicals from Around the World
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Medilink Pharmachem | India | |||
|---|---|---|---|---|
 | www.medilinkpharma.com | |||
 | +91 (79) 3007-0133 | |||
 | +91 (79) 4007-4175 | |||
 | exports@medilinkpharma.com | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Guizhou Jianye Yongrong Technology Co., Ltd. (Changzhou Huadong Chemical Research Institute) | China | |||
|---|---|---|---|---|
 | www.huadongchem.com | |||
 | +86 +13851563211 | |||
 | +86 (519) 8261-6386 | |||
 | sales@huadongchem.com | |||
 | WeChat: +13851563211 | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| abcr GmbH | Germany | |||
|---|---|---|---|---|
 | www.abcr.de | |||
 | +49 (721) 950-610 | |||
 | +49 (721) 950-6180 | |||
 | info@abcr.de | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Amine salt (ammonium salt) |
|---|---|
| Name | Benzyltripropylammonium chloride |
| Molecular Structure | 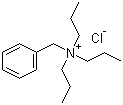 |
| Molecular Formula | C16H28ClN |
| Molecular Weight | 269.85 |
| CAS Registry Number | 5197-87-5 |
| EC Number | 625-674-9 |
| SMILES | CCC[N+](CCC)(CCC)CC1=CC=CC=C1.[Cl-] |
| Melting point | 190 °C (dec.) (Expl.) |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Benzyltripropylammonium chloride (BTPACl) is a quaternary ammonium salt that consists of a benzyl group attached to a nitrogen atom, which is also bonded to three propyl groups, with a chloride counterion. This compound belongs to a class of compounds known as alkylammonium salts, which are widely used in a variety of chemical applications. Quaternary ammonium salts, including BTPACl, are commonly studied for their surfactant properties, phase transfer catalytic activity, and as intermediates in organic synthesis. Benzyltripropylammonium chloride is typically synthesized through the reaction of benzyl chloride with tripropylamine under controlled conditions. In this process, the benzyl group is successfully attached to the nitrogen atom, and the chloride ion is incorporated as the counterion. This method of synthesis is well-documented and provides a high yield of BTPACl suitable for its various uses. One of the primary applications of benzyltripropylammonium chloride is in phase transfer catalysis. Phase transfer catalysis is a process in which a catalyst facilitates the transfer of a reactant between two immiscible solvents, typically an aqueous phase and an organic phase. In such reactions, BTPACl serves as a phase transfer agent by solubilizing ionic or polar reactants in organic solvents, allowing them to react with other substances in an organic phase. This ability to enable the transfer of reactants between different phases is especially useful for reactions involving nucleophilic substitutions or eliminations, where one reactant is in the aqueous phase and another is in the organic phase. In addition to its role in phase transfer catalysis, benzyltripropylammonium chloride is also employed in organic synthesis, particularly in reactions where the presence of a quaternary ammonium salt is beneficial. These reactions include the formation of various organoamines and amides, as well as the synthesis of other functionalized organic compounds. The presence of the benzyl and propyl groups provides specific steric and electronic properties that make BTPACl an effective reagent in certain synthetic pathways. Furthermore, BTPACl has been explored for its potential applications in materials science. It can be used in the preparation of surfactants and emulsifiers due to its amphiphilic nature, with both hydrophobic alkyl groups and a hydrophilic ammonium cation. This amphiphilic characteristic makes it useful for stabilizing emulsions and suspensions in various formulations, such as in cleaning products, personal care products, and other chemical formulations. Benzyltripropylammonium chloride also has applications in the preparation of ion-exchange resins and catalysts. Its ionic nature makes it suitable for use in processes that require the exchange of ions between a solid and a liquid phase. This is particularly useful in water treatment, where ion-exchange processes are employed to remove contaminants from water sources. Despite its usefulness in chemical processes, BTPACl must be handled with care. As a quaternary ammonium salt, it may have toxicological effects if improperly managed, and appropriate precautions, such as protective gloves and eyewear, should be taken when handling this compound. It is important to follow standard safety guidelines for working with chemicals, including using it in well-ventilated areas and avoiding direct contact with the skin or eyes. In summary, benzyltripropylammonium chloride is a quaternary ammonium salt that finds applications in phase transfer catalysis, organic synthesis, and materials science. Its ability to facilitate the transfer of reactants between different phases and its amphiphilic properties make it valuable in a variety of chemical processes. The continued use of BTPACl underscores its significance in both research and industrial applications, particularly in processes that involve organic chemistry and surfactant-based formulations. References 2024 Screening of neoteric solvents for gossypol extraction from cottonseed oil. Chemical Papers, 1, 1. DOI: 10.1007/s11696-024-03448-9 2022 (C16H28N)2SbCl5: A new lead-free zero-dimensional metal-halide hybrid with bright orange emission. Science China Materials, 1, 1. DOI: 10.1007/s40843-021-1937-y 2006 From 1,1-Dihaloalk-1-enes with a Hydrogen Atom in the α-Position. Science of Synthesis, 24, 130. DOI: 10.1055/sos-SD-024-00152 |
| Market Analysis Reports |