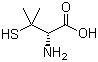
D-(-)-Penicillamine is a chelating agent and a modified form of the amino acid cysteine. It is primarily known for its medicinal uses in treating conditions such as rheumatoid arthritis, Wilson’s disease, and cystinuria. The compound is a derivative of penicillin, with its structure featuring a thiol group (-SH) that allows it to bind metal ions, making it effective in the treatment of diseases related to metal overload.
Penicillamine was first synthesized in the 1940s as part of the exploration of penicillin derivatives. Researchers discovered that modifying penicillin could yield compounds with new biological activities. D-(-)-Penicillamine is a stereoisomer of penicillamine, specifically the enantiomer that is active in clinical applications. It differs from the L-form of penicillamine in its optical activity, which influences its interaction with biological systems.
The most prominent application of D-(-)-Penicillamine is in the treatment of Wilson’s disease, a rare genetic disorder in which copper accumulates in the body, leading to liver damage and neurological impairment. D-(-)-Penicillamine works by forming stable complexes with copper ions, which are then excreted in the urine, thereby reducing the harmful copper buildup. This chelation therapy has significantly improved the quality of life for patients with Wilson’s disease and has been used since the 1950s. The drug is often prescribed for long-term use, and its effectiveness has been well-documented in clinical studies.
In addition to Wilson's disease, D-(-)-Penicillamine is used in the management of cystinuria, a hereditary disorder characterized by the formation of cystine stones in the kidneys due to the inability to properly metabolize cysteine. D-(-)-Penicillamine helps to prevent the formation of these stones by binding to cysteine, reducing its tendency to form insoluble crystals. It is also used in the treatment of rheumatoid arthritis, where it functions as a disease-modifying antirheumatic drug (DMARD). In rheumatoid arthritis, D-(-)-Penicillamine is thought to work by modulating the immune system and reducing inflammation, although its precise mechanism of action in this context remains not fully understood.
The compound has also been explored for its potential in treating other conditions that involve metal toxicity, including lead poisoning, although its use in these areas is less common. D-(-)-Penicillamine’s ability to chelate metals makes it a versatile tool in treating conditions associated with heavy metal accumulation.
While D-(-)-Penicillamine is generally effective in treating these conditions, it can have side effects. Common adverse effects include gastrointestinal symptoms like nausea, vomiting, and loss of appetite. More serious side effects can include skin rashes, proteinuria, and, in rare cases, autoimmune reactions or hematologic disorders like agranulocytosis or thrombocytopenia. Due to these potential side effects, its use is carefully monitored, and patients often undergo regular blood tests to ensure their safety during treatment.
Despite these risks, D-(-)-Penicillamine remains a critical drug in the management of metal-related disorders. It is typically prescribed when other therapies are ineffective or inappropriate, and it has been found to be especially beneficial in preventing the severe, life-threatening effects of copper toxicity in Wilson’s disease. Its role in managing cystinuria and rheumatoid arthritis also makes it a valuable tool in chronic disease management.
In conclusion, D-(-)-Penicillamine is a potent chelating agent with established applications in the treatment of Wilson’s disease, cystinuria, and rheumatoid arthritis. It functions by binding to metal ions, particularly copper, and facilitating their excretion from the body. Although its use can be associated with side effects, its therapeutic benefits for patients with metal overload disorders have been well-documented and remain a cornerstone of treatment for these conditions.
References
1990. The comparative efficacy and toxicity of second-line drugs in rheumatoid arthritis. Results of two metaanalyses. Arthritis and Rheumatism, 33(10).
DOI: 10.1002/art.1780331001
1991. Prognosis of Wilsonian chronic active hepatitis. Gastroenterology, 100(3).
DOI: 10.1016/0016-5085(91)80023-3
1991. Autoantibodies in D-penicillamine-induced myasthenia gravis: A comparison with idiopathic myasthenia and rheumatoid arthritis. Clinical Immunology and Immunopathology, 58(3).
DOI: 10.1016/0090-1229(91)90123-r
|


























































 GHS07 Warning Details
GHS07 Warning Details