O-Methylisourea hemisulfate is an important chemical compound widely used in organic synthesis and biochemical research. It consists of an O-methylisourea cation paired with a hemisulfate anion, making it a stable and soluble form of O-methylisourea. The compound is primarily recognized for its role in methylation reactions and its application in the modification of biomolecules.
The discovery of O-methylisourea hemisulfate is closely linked to studies on guanidine derivatives and their reactivity in organic synthesis. Researchers have long investigated the role of isourea derivatives in alkylation and methylation processes, leading to the development of stable salt forms to enhance their reactivity and handling. The hemisulfate form was introduced to improve solubility and facilitate its use in aqueous systems.
One of the key applications of O-methylisourea hemisulfate is in the synthesis of methylated nucleobases and amino acids. It serves as a methylating agent in reactions where selective modification of nitrogen-containing functional groups is required. In biochemistry, it has been utilized for modifying proteins and peptides, influencing their structure and function. The compound is also employed in pharmaceutical research, particularly in the development of enzyme inhibitors and bioactive small molecules.
Beyond biochemical applications, O-methylisourea hemisulfate is used in material sciences for the modification of polymers and coatings. Its ability to introduce methyl functional groups allows for the fine-tuning of surface properties and chemical reactivity. Additionally, its controlled reactivity makes it valuable in the study of methylation mechanisms in biological and chemical systems.
As research continues, the role of O-methylisourea hemisulfate in synthetic and medicinal chemistry is expanding. New methodologies are being developed to enhance its efficiency in targeted modifications, making it a crucial tool for advancing chemical and biological sciences.
References
2022. Growing Impact of Ionic Liquids in Heterocyclic Chemistry. Green Chemistry.
DOI: 10.1007/978-981-19-2734-8_4
2002. A New Pathway to Aspartic Acid from Urea and Maleic Acid Affected by Ultraviolet Light. Origins of life and evolution of the biosphere : the journal of the International Society for the Study of the Origin of Life, 32(2).
DOI: 10.1023/a:1016067303182
|



























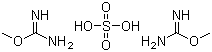

 GHS07;GHS08 Warning Details
GHS07;GHS08 Warning Details