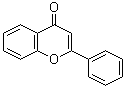
Flavonoids are naturally occurring organic compounds belonging to the class of flavonoids that are recognized for their diverse uses and biological significance. Flavonoids have the molecular formula C15H10O2 and were first isolated in 1930 by chemist J.C. de Jonge from the flowers of Pseudocydonia sinensis. Their structure is characterized by a benzene ring fused to a pyrone ring, forming a scaffold that is essential for their function. This structure, with its unique aromatic rings connected by a three-carbon chain, plays a key role in the application of this compound in various fields.
In the medical field, flavonoids have attracted much attention due to their impressive biological activities. One of their main roles is as an antioxidant. Flavonoids have the ability to scavenge free radicals, thereby protecting cells from oxidative stress and damage. This antioxidant property helps prevent chronic diseases, including cancer and cardiovascular disease. In addition to their role as antioxidants, flavonoids also have anti-inflammatory, antiviral, and antiallergic effects, which are essential for exploring new therapeutic avenues. In particular, flavonoids and their derivatives have shown good promise in modulating the activity of enzymes associated with cancer cell growth and proliferation. Ongoing research aims to exploit these properties to develop new treatments that could offer improved efficacy and safety compared to existing treatments.
The application of flavonoids extends beyond medicine to agriculture. The compounds play an important role as natural plant protectants. Flavonoid derivatives help to enhance plant resistance to various pests and diseases. This property reduces the reliance on synthetic pesticides and is in line with sustainable agricultural practices. In addition, flavonoid-based compounds are used to develop natural herbicides and fungicides, providing an environmentally friendly alternative to traditional chemical agents. These applications highlight the role of flavonoids in promoting sustainable agriculture and reducing the ecological impact of crop protection.
In addition to its medicinal and agricultural uses, flavonoids are also valuable in materials science. Its bright color and ability to form complexes with metal ions make it useful in making dyes and pigments. The photophysical properties of the compound have prompted the exploration of its use in light-emitting devices and sensors, and it helps in the development of advanced materials with special functionalities. This versatility in materials science highlights the wide range of uses of flavonoids in different industrial applications.
Flavonoids are generally considered safe in terms of environmental and safety considerations, especially considering its natural occurrence and biodegradability. Compared to synthetic chemicals, flavonoids are less likely to pose long-term environmental hazards. However, as with any chemical, proper handling and application are essential to ensure safety and effectiveness, especially when used in concentrated form for industrial purposes.
References
Benjamin Willy, Walter FrankX-Ray structure analyses. and Thomas J. J. Müller. Microwave-assisted three-component coupling-addition-SAr (CASNAR) sequences to annelated 4H-thiopyran-4-ones, Org. Biomol. Chem., 2010, 8, 90.
|




















 GHS07 Warning Details
GHS07 Warning Details