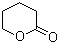
Delta-valerolactone (DVL) is a cyclic ester known for its diverse applications in chemical synthesis and industry. Its discovery and subsequent development have underscored its versatility as a chemical intermediate with significant industrial and commercial relevance.
Discovered in the mid-20th century, delta-valerolactone is a five-membered lactone derived from the condensation of gamma-hydroxybutyric acid (GHB). The synthesis of DVL involves the cyclization of 1,4-pentadiol or similar precursors, which forms the lactone ring structure. This process was initially explored to create compounds with specific reactivity profiles and potential uses in various chemical reactions.
In industrial applications, delta-valerolactone serves as an important intermediate in the production of various chemicals. One of its primary uses is in the synthesis of polyesters and resins. The lactone ring of DVL can be polymerized to form polymers with desirable mechanical properties and stability. These polymers find applications in coatings, adhesives, and other materials where durability and performance are critical.
Delta-valerolactone is also used as a solvent in chemical processes. Its ability to dissolve a range of organic compounds makes it valuable in the formulation of industrial solvents and cleaning agents. The solvent properties of DVL are particularly useful in formulations where a high degree of solvency and low toxicity are desired.
In the pharmaceutical industry, DVL is employed as a precursor in the synthesis of various drugs and therapeutic agents. Its ability to participate in ring-opening polymerization reactions allows for the creation of biologically active compounds with potential therapeutic benefits. Research into the applications of DVL in drug synthesis continues to explore its role in developing new medications and treatments.
Another notable application of delta-valerolactone is in the production of biodegradable materials. The polymerization of DVL can lead to the formation of biodegradable polymers, which are of increasing interest due to environmental concerns about plastic waste. These materials offer potential solutions for reducing the ecological impact of traditional plastics.
Despite its many applications, delta-valerolactone is relatively less well-known compared to other lactones and chemicals. However, its role in industrial chemistry and its potential for use in new materials and pharmaceuticals highlight its significance in various fields.
In summary, delta-valerolactone is a valuable chemical compound with a range of applications in industrial and pharmaceutical settings. Its role as an intermediate in polymer synthesis, solvent use, and the development of biodegradable materials demonstrates its versatility and importance in modern chemistry.
References
2024. New sustainable polymers with on-demand depolymerization property. Science China Chemistry.
DOI: 10.1007/s11426-024-2167-9
2024. Controllable Ring-opening Polymerization of δ-Valerolactone Catalyzed by Quinolinyl-Urea/MTBD Systems. Chinese Journal of Polymer Science.
DOI: 10.1007/s10118-024-3127-3
2023. Preparation of Chemically Recyclable Poly(ether-alt-ester) by the Ring Opening Polymerization of Cyclic Monomers Synthesized by Coupling Glycolide and Epoxides. Chinese Journal of Polymer Science, 41(11).
DOI: 10.1007/s10118-023-3040-1
|







































 GHS05 Danger Details
GHS05 Danger Details