Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shanghai Rory Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rorychem.com | |||
 | +86 (21) 5070-2305 | |||
 | +86 (21) 5070-4994 | |||
 | rorychem@hotmail.com 13701863355@139.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Quzhou HuntBio Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huntchem.com | |||
 | +86 (571) 6406-7827 +86 13868119375 | |||
 | +86 (571) 6479-8288 | |||
 | sales@huntchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Ich Biofarm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ichemie.com | |||
 | +86 (571) 2818-6870 | |||
 | +86 (571) 8993-9479 | |||
 | specialchem1@ichemie.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Frontier Specialty Chemicals | USA | |||
|---|---|---|---|---|
 | www.frontierspecialtychemicals.com | |||
 | +1 (435) 753-1901 | |||
 | +1 (435) 753-6731 | |||
 | sales@frontierspecialtychemicals.com | |||
| Chemical manufacturer since 1975 | ||||
| Heterocyclics Inc. | USA | |||
|---|---|---|---|---|
 | www.heterocyclics.com | |||
 | +1 (732) 444-6336 | |||
 | +1 (732) 444-4326 | |||
 | info@heterocyclics.com jagathreddy@usa.net | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Synthonix, Inc. | USA | |||
|---|---|---|---|---|
 | www.synthonix.com | |||
 | +1 (919) 875-9277 | |||
 | +1 (919) 875-9601 | |||
 | info@synthonix.com | |||
| Chemical manufacturer | ||||
| Paragos e. K. | Germany | |||
|---|---|---|---|---|
 | www.paragos.com | |||
 | +49 (2330) 807-9751 | |||
 | +49 (2330) 807-9752 | |||
 | sales@paragos.de | |||
| Chemical manufacturer since 2001 | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridazine |
|---|---|
| Name | 6-Chloropyridazin-3-amine |
| Synonyms | 3-Amino-6-chloropyridazine |
| Molecular Structure | 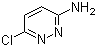 |
| Molecular Formula | C4H4ClN3 |
| Molecular Weight | 129.55 |
| CAS Registry Number | 5469-69-2 |
| EC Number | 226-796-1 |
| SMILES | C1=CC(=NN=C1N)Cl |
| Density | 1.4±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 215 - 220 °C (Expl.) |
| Boiling point | 363.2±22.0 °C 760 mmHg (Calc.)* |
| Flash point | 173.4±22.3 °C (Calc.)* |
| Index of refraction | 1.618 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
6-Chloropyridazin-3-amine is a chemical compound belonging to the class of pyridazine derivatives. It consists of a pyridazine ring, which is a six-membered heterocyclic structure containing two nitrogen atoms, with a chlorine atom attached to the 6-position and an amine group (-NH2) at the 3-position. This structural arrangement makes it a functionalized pyridazine derivative with potential applications in various fields, including organic synthesis, pharmaceuticals, and materials science. The synthesis of 6-chloropyridazin-3-amine typically involves halogenation of pyridazine, followed by selective introduction of the amine group. The chlorination step is often carried out using reagents such as chlorine gas or a chlorinating agent, which introduce the chlorine atom at the desired position. The amine group can be introduced through standard amination techniques, utilizing either a direct reaction with an appropriate amine or through the use of reagents that facilitate nucleophilic substitution at the 3-position of the pyridazine ring. This compound is of particular interest in medicinal chemistry due to the presence of both a halogen (chlorine) and an amine group. The combination of these functional groups can impart unique reactivity and biological activity to the molecule. The amine group, being a nucleophilic site, can participate in a variety of chemical reactions, including the formation of salts, coordination complexes, or other derivatives. Meanwhile, the chlorine atom can influence the compound's lipophilicity, reactivity, and interaction with biological targets. 6-Chloropyridazin-3-amine has been studied in the context of drug discovery, particularly for its potential as a building block in the synthesis of biologically active compounds. Pyridazine derivatives are known to exhibit a range of pharmacological properties, including antimicrobial, anti-inflammatory, and anticancer activities. The presence of the amine group enhances the compound's ability to engage in interactions with various biological molecules, such as enzymes or receptors, which may explain its potential therapeutic applications. Moreover, this compound could be a useful intermediate in the synthesis of more complex pyridazine-based pharmaceuticals or agrochemicals. By modifying the functional groups on the pyridazine ring, it is possible to tailor the compound's properties for specific applications. The ability to easily functionalize the amine and halogen groups provides synthetic flexibility, which is beneficial in designing compounds with improved potency, selectivity, and pharmacokinetic properties. Additionally, 6-chloropyridazin-3-amine may find applications in materials science. Pyridazine derivatives are sometimes used in the development of new materials with specific electronic, optical, or conductive properties. By incorporating functional groups such as amines and halogens, the chemical properties of these materials can be tuned to suit specific industrial needs, such as in the creation of polymers, coatings, or sensors. In summary, 6-chloropyridazin-3-amine is a versatile and potentially valuable compound in both synthetic and pharmaceutical chemistry. Its ability to undergo various chemical transformations, along with its presence in biologically active molecules, makes it a promising candidate for further study in drug development and materials science. References 2006. Palladium-Catalysed Cross-Coupling Reactions on Pyridazine Moieties. Synlett, 2006(17). DOI: 10.1055/s-2006-951528 2011. First Synthesis of 3-Amino-2-arylimidazo[1,2-b]pyridazines by Groebke-Blackburn Reaction. Synlett, 2011(10). DOI: 10.1055/s-0030-1260940 2023. Synthesis, Crystal Structure, and DFT Study of Ethyl 6-Chloro-3-fluoroimidazo[1,2-b]pyridazine-2-carboxylate. Russian Journal of General Chemistry, 93(5). DOI: 10.1134/s1070363223050201 |
| Market Analysis Reports |