Online Database of Chemicals from Around the World
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Zolanbio Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zolanbio.com | |||
 | +86 13071211027 | |||
 | helen@zolanbio.cn | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | Helenien |
| Synonyms | Xantofyl palmitate; beta-Carotene-4,4'-diol dipalmitate |
| Molecular Structure | 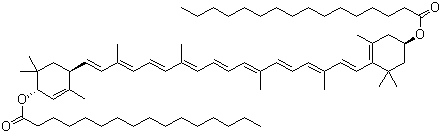 |
| Molecular Formula | C72H116O4 |
| Molecular Weight | 1045.69 |
| CAS Registry Number | 547-17-1 |
| EC Number | 208-916-4 |
| SMILES | CCCCCCCCCCCCCCCC(=O)O[C@@H]1CC(=C(C(C1)(C)C)/C=C/C(=C/C=C/C(=C/C=C/C=C(C)/C=C/C=C(C)/C=C/[C@H]2C(=C[C@@H](CC2(C)C)OC(=O)CCCCCCCCCCCCCCC)C)/C)/C)C |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 956.4±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 462.1±32.7 °C (Calc.)* |
| Index of refraction | 1.524 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Helenien, also known as lutein dipalmitate (or β,ε-carotene-3,3′-diol dipalmitate), is a carotenoid ester belonging to the xanthophyll class of pigments. Its molecular formula is C72H116O4 and its molecular weight is approximately 1045.7 g mol−1. The compound is most commonly extracted from marigold flowers (Tagetes erecta), where lutein esters (including lutein dipalmitate) accumulate in the petals and serve as major colouring agents. Helenien was identified in the context of carotenoid studies and pigment analysis of marigold and other yellow-flowered plants. Researchers noted that marigold petals contained a significant proportion of esterified lutein (primarily palmitate esters) together with free lutein, and analytical separation revealed the dipalmitate ester form which was then referred to as “helenien”. The term is rooted in older naming conventions for xanthophylls derived from plant sources. As a pigment it contributed to the characteristic orange-yellow coloration of marigold petals. The core structure of Helenien is derived from lutein, a dihydroxycarotenoid with two terminal rings (β and ε) and a conjugated polyene chain. In Helenien, each of the two hydroxyl groups at positions 3 and 3′ of the lutein backbone is esterified with a palmitoyl (hexadecanoyl) fatty acid. This esterification alters its physicochemical properties: Helenien is more lipophilic, less polar than free lutein, and exhibits different solubility and chromatographic behavior. The ester linkage can be hydrolysed under suitable conditions to release free lutein and palmitic acid. In terms of application, Helenien is primarily found in the food coloring, dietary supplement and pigment industries. Because it is a major component of commercially-available marigold extracts (used for lutein production), Helenien serves as a marker compound for the quality control of such extracts. Upon ingestion, the esterified lutein forms (including Helenien) are hydrolysed in the intestine to free lutein, which can then be absorbed and contribute to macular pigment in the human retina. Thus, although Helenien itself may not be the active molecule in vivo, it functions as a delivery or storage form for lutein in botanical preparations. Beyond human nutrition, Helenien (lutein dipalmitate) is also studied for its antioxidant and photoprotective properties when incorporated into lipid-based formulations, and for use in colour-stable pigments for food and feed applications. Regulatory evaluations by agencies such as the Joint FAO/WHO Expert Committee on Food Additives (JECFA) list lutein esters extracted from Tagetes (including Helenien) under “Tagetes extract (colour)” and provide compositional specifications for lutein esters. According to JECFA monographs, the definition of Helenien is “lutein dipalmitate; β,ε-carotene-3,3′-diol dipalmitate”. From a practical standpoint, when handling Helenien as a reference standard or ingredient, note that it is highly lipophilic, typically a dark yellow-brown liquid or solid at room temperature, insoluble in water and soluble in non-polar organic solvents (e.g., hexane). The ester linkages confer increased stability in fatty matrices, but the material should still be protected from light, oxygen and heat to avoid degradation of the carotenoid polyene chain. Overall, Helenien exemplifies the esterified xanthophyll pigment forms found in nature; its role as a storage form of lutein in marigold petals, its use in food colouring and supplements, and its relevance for human nutrition and pigment chemistry make it a compound of continuing interest in both research and industrial contexts. References FAO/WHO (2001) TAGETES EXTRACT. Evaluation of certain food additives. Thirty-first report of the Joint FAO/WHO Expert Committee on Food Additives. WHO Technical Report Series No. 901. FAO, Rome. El-Sayed MA, Hinner MP, Tomas JA, Abdul-Ghani A & Kassem TA (2015) Composition of lutein ester regioisomers in marigold flower, dietary supplement, and herbal tea. Journal of Agricultural and Food Chemistry 63(44) 9740–9746. DOI: 10.1021/acs.jafc.5b04107 Smith MA & Johnson EJ (2016) Role of lutein and its esters in human nutrition and vision. Nutrition Reviews 74(9) 706–720. DOI: 10.1093/nutrit/nuw023 |
| Market Analysis Reports |