Online Database of Chemicals from Around the World
| Shijiazhuang Dowell Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dowechem.com | |||
 | +86 (311) 8980-5679 +86 13463963265 | |||
 | +86 (311) 8526-2695 | |||
 | sell@dowechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shijiazhuang C-element Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.c-elementbiotech.com | |||
 | +86 18033710157 | |||
 | Lisa@C-elementBiotech.com | |||
 | QQ Chat | |||
 | WeChat: 18033710157 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Interchim | France | |||
|---|---|---|---|---|
 | www.interchim.com | |||
 | +33 (4) 7003-7301 | |||
 | interfine@mail.interchim.fr | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| abcr GmbH | Germany | |||
|---|---|---|---|---|
 | www.abcr.de | |||
 | +49 (721) 950-610 | |||
 | +49 (721) 950-6180 | |||
 | info@abcr.de | |||
| Chemical manufacturer | ||||
| MP Biomedicals LLC. | USA | |||
|---|---|---|---|---|
 | www.mpbio.com | |||
 | +1 (440) 337-1200 | |||
 | +1 (440) 337-1180 | |||
 | inside@mpbio.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Plant extracts |
|---|---|
| Name | 3-Butylidenephthalide |
| Synonyms | (3Z)-3-butylidene-2-benzofuran-1-one |
| Molecular Structure | 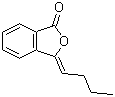 |
| Molecular Formula | C12H12O2 |
| Molecular Weight | 188.22 |
| CAS Registry Number | 551-08-6 |
| EC Number | 208-991-3 |
| FEMA | 3333 |
| SMILES | CCC/C=C1/C2=CC=CC=C2C(=O)O1 |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.103 g/mL (Expl.) |
|---|---|
| Index of Refraction | 1.631, Calc.*, 10577 (Expl.) |
| Boiling Point | 312.0±35.0 °C (760 mmHg), Calc.*, 330-344.9 °C (Expl.) |
| Flash Point | 127.9±23.4 °C, Calc.*, 132 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
3-Butylidenephthalide is a chemical compound known for its structure and distinct reactivity, falling under the class of phthalide derivatives. It is a synthetic organic compound with significant interest in chemical synthesis and potential applications in pharmaceuticals and materials science. The compound is characterized by the phthalide ring structure, with a butylidene group at the 3-position, which influences its chemical properties and reactivity. The discovery of 3-butylidenephthalide can be traced to efforts aimed at modifying phthalide derivatives for improved chemical stability and reactivity in various synthetic applications. Phthalides themselves are a class of compounds that have been studied for their bioactive properties and their potential use in medicinal chemistry. The butylidene substitution at the 3-position of the phthalide ring offers enhanced stability and opens up further avenues for chemical modifications, which are important in designing novel compounds. 3-Butylidenephthalide has found applications in organic synthesis, where it is used as an intermediate in the preparation of more complex organic molecules. Its reactivity is utilized in several organic reactions, including nucleophilic substitutions and cyclization processes, which can lead to the formation of larger, more complex structures. This makes it valuable for the synthesis of various organic compounds used in pharmaceuticals, agrochemicals, and other industrial chemicals. In the pharmaceutical industry, derivatives of phthalide compounds, including 3-butylidenephthalide, have been studied for their biological activities. While the specific biological applications of 3-butylidenephthalide are still an area of ongoing research, similar compounds in the phthalide family have been shown to possess a range of pharmacological activities, including anti-inflammatory, antimicrobial, and anticancer properties. The modification of the phthalide ring with a butylidene group is likely to affect the compound’s interactions with biological targets, thus potentially contributing to the development of new therapeutic agents. The compound's role in materials science is also of interest, particularly in the development of advanced materials with specific properties. For example, the unique chemical structure of 3-butylidenephthalide may provide useful building blocks for the creation of novel polymers or other materials with tailored characteristics, such as improved solubility, stability, or conductivity. In summary, 3-butylidenephthalide is an important intermediate in organic synthesis, with potential applications in pharmaceuticals and materials science. While research is ongoing to fully explore its bioactive properties and industrial uses, its role as a versatile building block in chemical synthesis is well established. References 2023. (Z)-3-Butylidenephthalide from Angelica sinensis (Dong quai) ameliorates memory impairment and neurological deficit induced by scopolamine in mice through the regulation of the BDNF/TrkB/CREB signaling pathway. Phytotherapy Research, 37(8). DOI: 10.1002/ptr.7818 2021. RIFM fragrance ingredient safety assessment, 3-butylidenephthalide, CAS Registry Number 551-08-6. Food and Chemical Toxicology: An International Journal Published for the British Industrial Biological Research Association, 155. DOI: 10.1016/j.fct.2021.112490 2015. (Z)-3-Butylidenephthalide Protects Against Concanavalin A-induced Hepatic Injury in Mice Through the Toll-Like Receptor 4 Signaling Pathway. Planta Medica, 81(16). DOI: 10.1055/s-0035-1558098 |
| Market Analysis Reports |