Online Database of Chemicals from Around the World
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Suzhou Biosyntech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biosyntech-suzhou.com | |||
 | +86 13921151340 +86 (512) 6300-1269 | |||
 | sales2@biosyntech-suzhou.com sales@biosyntech-suzhou.com | |||
 | QQ Chat | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | (2S,3S,4R)-2-Amino-1,3,4-octadecanetriol |
| Synonyms | Phytosphingosine; (+)-D-ribo-Phytosphingosine; 4-D-Hydroxysphinganine; 4D-Hydroxysphinganine; C18-Phytosphingosine; D-ribo-1,3,4-Trihydroxy-2-aminooctadecane |
| Molecular Structure | 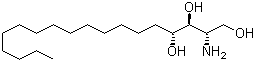 |
| Molecular Formula | C18H39NO3 |
| Molecular Weight | 317.51 |
| CAS Registry Number | 554-62-1 |
| EC Number | 439-210-6 |
| SMILES | CCCCCCCCCCCCCC[C@H]([C@H]([C@H](CO)N)O)O |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 102 °C (Expl.) |
| Boiling point | 483.7±40.0 °C 760 mmHg (Calc.)* |
| Flash point | 246.4±27.3 °C (Calc.)* |
| Index of refraction | 1.49 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS09 Danger Details GHS05;GHS09 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H318-H400-H410 Details | ||||||||||||||||
| Safety Statements | P264+P265-P273-P280-P305+P354+P338-P317-P391-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
(2S,3S,4R)-2-Amino-1,3,4-octadecanetriol is a stereochemically defined aliphatic amino alcohol, classified within the broader family of sphingoid bases, which are long-chain amino alcohols that serve as fundamental structural units of sphingolipids. These lipids are essential components of cell membranes, particularly in neural tissue, and play critical roles in signal transduction, cell recognition, and apoptosis. The compound's molecular structure features an 18-carbon saturated hydrocarbon chain with three distinct functional groups located on carbons 1, 3, and 4 as hydroxyl groups and on carbon 2 as a primary amine, each with specific stereochemistry. The discovery of such long-chain bases traces back to the early biochemical studies on sphingolipids in the 19th and 20th centuries. The term "sphingosine" was first introduced in the 1880s due to the mysterious nature of the lipid extract from brain tissue. Later advancements in organic and analytical chemistry enabled the identification of related sphingoid bases such as phytosphingosine and dihydrosphingosine, which bear similarity in backbone structure to (2S,3S,4R)-2-amino-1,3,4-octadecanetriol. While not as commonly encountered in natural systems as phytosphingosine or sphingosine itself, (2S,3S,4R)-2-amino-1,3,4-octadecanetriol represents a synthetic or less abundant natural analog of these key bioactive molecules. This compound's structural configuration suggests it could be derived biosynthetically from saturated fatty acid precursors through enzymatic processes involving serine palmitoyltransferase and subsequent reductions and hydroxylations. The stereochemical arrangement is crucial, as biological activity and incorporation into sphingolipids are highly dependent on the precise spatial orientation of the functional groups. The (2S,3S,4R) configuration allows for specific interactions with enzymes such as ceramide synthases, sphingosine kinases, and glycosyltransferases, potentially leading to incorporation into complex lipids or serving as intermediates in metabolic pathways. In application, such compounds are studied in the context of dermatology, cancer biology, and neurobiology. Synthetic analogs of naturally occurring sphingoid bases have been utilized in skincare formulations due to their ability to restore the skin barrier, enhance hydration, and modulate inflammatory responses. The amphipathic nature of (2S,3S,4R)-2-amino-1,3,4-octadecanetriol enables it to integrate into lipid bilayers, influencing membrane microdomains known as lipid rafts. These rafts are critical for organizing membrane proteins and mediating signaling cascades, particularly in immune and neuronal cells. In addition, research into analogs like (2S,3S,4R)-2-amino-1,3,4-octadecanetriol has extended to anticancer strategies. Certain sphingoid bases and their derivatives exhibit cytotoxic effects on cancer cells through mechanisms involving ceramide accumulation, mitochondrial dysfunction, and activation of apoptotic pathways. The presence of the amino group and three hydroxyl groups allows for multiple metabolic conversions, including phosphorylation, acylation, and glycosylation, leading to biologically active molecules that can either promote or inhibit cell proliferation depending on cellular context. Synthetic routes to produce (2S,3S,4R)-2-amino-1,3,4-octadecanetriol involve enantioselective synthesis or stereocontrolled reduction of suitable ketone or epoxide intermediates, often starting from fatty acid precursors. Protection-deprotection strategies and chiral auxiliary techniques are commonly employed to ensure the correct stereochemical outcome. The compound's physical characteristics, such as high melting point and limited aqueous solubility, are consistent with its long aliphatic chain and multiple polar functional groups. Overall, (2S,3S,4R)-2-amino-1,3,4-octadecanetriol represents a valuable molecule in biochemical research and pharmaceutical development. It serves as both a model compound for studying sphingolipid metabolism and a potential building block for novel therapeutic agents targeting lipid-mediated pathways in health and disease. References 1957. The phosphatides and glycolipids. Stoffwechselphysiologie der Fette und Fettähnlicher Stoffe / The Metabolism of Fats and Related Compounds. DOI: 10.1007/978-3-642-94704-9_13 2024. A ‘Chiron’ approach to novel phytosphingosine mimetics based on a cascade [3,3]-sigmatropic rearrangement. Carbohydrate Research, 541. DOI: 10.1016/j.carres.2024.109158 2024. Anti-inflammatory effects of phytosphingosine-regulated cytokines and NF-kB and MAPK mechanism. Cellular and Molecular Biology, 70(9). DOI: 10.14715/cmb/2024.70.9.3 |
| Market Analysis Reports |