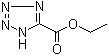
Ethyl tetrazole-5-carboxylate is an organic compound that belongs to the tetrazole class of chemicals, characterized by a five-membered ring containing four nitrogen atoms and one carbon atom. This compound has been primarily used in the synthesis of tetrazole derivatives and has applications in various fields, including medicinal chemistry, materials science, and agricultural chemistry.
The discovery of ethyl tetrazole-5-carboxylate is linked to the broader study and development of tetrazole-based compounds, which are recognized for their diverse chemical reactivity and biological activities. Tetrazole derivatives, including this compound, have been investigated for their potential as bioactive agents, owing to the reactive nature of the tetrazole ring that can form coordination complexes with metals, interact with biological targets, and undergo nucleophilic substitutions.
Ethyl tetrazole-5-carboxylate is most commonly utilized as a key intermediate in the synthesis of other tetrazole derivatives, particularly in the preparation of bioactive molecules. The ethyl ester group at the 5-position of the tetrazole ring enhances its solubility and reactivity, making it a useful building block for the creation of more complex structures. This compound has been employed in the design and development of pharmaceutical agents, including those with potential antifungal, antiviral, and anticancer properties.
In the field of agricultural chemistry, ethyl tetrazole-5-carboxylate has found application as an intermediate in the synthesis of agrochemicals, such as herbicides and insecticides. Its ability to participate in various chemical reactions, including esterification and nucleophilic substitution, allows for the modification of the tetrazole ring to produce compounds with specific biological activities aimed at pest control.
Beyond its use in pharmaceuticals and agrochemicals, ethyl tetrazole-5-carboxylate has been explored for its potential applications in materials science. The reactivity of the tetrazole ring has led to the development of coordination polymers and metal-organic frameworks (MOFs) that exhibit interesting properties, including catalytic activity, gas storage capabilities, and electronic conductivity. As such, tetrazole-based compounds are of growing interest for use in advanced materials.
In conclusion, ethyl tetrazole-5-carboxylate is an important intermediate in the synthesis of a range of bioactive and functional materials. Its role in medicinal chemistry, agricultural applications, and materials science highlights its versatility and the continued interest in tetrazole derivatives for a variety of industrial and research purposes.
References
2013. Efficient Transformation of Inactive Nitriles into 5-Substituted 1H-Tetrazoles Using Microwave Irradiation and Their Applications. Synthesis.
DOI: 10.1055/s-0032-1318476
2009. A Simple, Advantageous Synthesis of 5-Substituted 1H-Tetrazoles¹. Synlett.
DOI: 10.1055/s-0029-1219150
1987. Derivatives of 1-hydroxytetrazole-5-carboxylic acid. Bulletin of the Academy of Sciences of the USSR, Division of chemical science.
DOI: 10.1007/bf00957315
|































 GHS07 Warning Details
GHS07 Warning Details