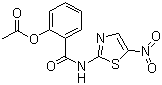
Nitazoxanide is a broad-spectrum antiparasitic and antiviral agent that belongs to the thiazolide class of compounds. Chemically, it is an N-(5-nitro-2-thiazolyl)amide derivative, with a 2-acetoxyphenyl substituent. The compound was first synthesized in the late 1970s as part of research into novel antiprotozoal agents. Its discovery was driven by the need for effective treatment against intestinal parasites such as Giardia lamblia and Cryptosporidium parvum, which were not adequately controlled by existing drugs. Early studies demonstrated its high efficacy in vitro and in vivo against these protozoa, leading to further clinical development.
The pharmacological activity of nitazoxanide stems from its interference with the pyruvate:ferredoxin oxidoreductase (PFOR) enzyme-dependent electron transfer reaction, which is essential for anaerobic energy metabolism in protozoa and some anaerobic bacteria. By inhibiting this pathway, nitazoxanide disrupts ATP production, leading to the death of the parasitic organism. Its mechanism is selective for anaerobic pathogens because human cells utilize different enzymatic pathways for energy metabolism. In addition to its antiparasitic activity, nitazoxanide has demonstrated broad antiviral properties, showing inhibitory effects against influenza viruses, rotaviruses, hepatitis B and C viruses, and certain coronaviruses in preclinical studies.
Nitazoxanide is a prodrug that undergoes rapid hydrolysis to its active metabolite, tizoxanide, after oral administration. Tizoxanide is the primary active species responsible for the pharmacological effects. The compound exhibits favorable pharmacokinetic properties, including rapid absorption, extensive tissue distribution, and a short plasma half-life, necessitating twice-daily dosing in clinical use. Its oral bioavailability and tolerability make it suitable for treatment in both adults and pediatric populations.
Clinically, nitazoxanide is widely used to treat intestinal protozoal infections, particularly giardiasis and cryptosporidiosis. It has also been evaluated in combination therapies for viral infections, including hepatitis C, where it enhances antiviral efficacy. Its broad spectrum of activity and low incidence of resistance have contributed to its adoption in regions with high prevalence of protozoal infections. Furthermore, nitazoxanide has been investigated for potential use against emerging viral pathogens due to its host-targeted mechanism, which reduces the likelihood of resistance development.
From a chemical synthesis perspective, nitazoxanide is prepared by acylation of 2-acetoxybenzoyl chloride with 2-amino-5-nitrothiazole. The process requires careful control of reaction conditions to ensure high yield and purity. The resulting compound is typically formulated as an oral suspension or tablet for therapeutic use. Its chemical stability, ease of formulation, and water solubility contribute to its practical utility as an oral medication.
In addition to its direct therapeutic applications, nitazoxanide serves as a model compound for the development of other thiazolide derivatives with improved antiviral or antiparasitic profiles. Its dual activity against protozoal and viral pathogens exemplifies the potential for repurposing existing drugs for multiple indications. Overall, nitazoxanide is a significant pharmaceutical agent with broad clinical applications, well-established pharmacology, and versatile utility in both research and therapeutic contexts.
|















































 GHS07 Warning Details
GHS07 Warning Details