Online Database of Chemicals from Around the World
| ChemPacific Corp | USA | |||
|---|---|---|---|---|
 | www.chempacific.com | |||
 | +1 (410) 633-5771 | |||
 | +1 (410) 633-5808 | |||
 | sales@chempacific.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Alpha Chemical (Shijiazhuang) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dzalpha.com | |||
 | +86 (311) 8565-2680 | |||
 | +86 (311) 8565-2789 | |||
 | market@dzalpha.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| H. W. Sands | USA | |||
|---|---|---|---|---|
 | www.hwsands.com | |||
 | +1 (561) 743-8090 | |||
 | +1 (561) 743-4088 | |||
 | customerservice@hwsands.com | |||
| Chemical manufacturer since 1983 | ||||
| OChem Incorporation | USA | |||
|---|---|---|---|---|
 | www.ocheminc.com | |||
 | +1 (847) 403-7044 | |||
 | +1 (847) 298-5008 | |||
 | sales@ocheminc.com | |||
| Chemical manufacturer | ||||
| Spectrum Chemical Mfg. Corp. | USA | |||
|---|---|---|---|---|
 | www.spectrumchemical.com | |||
 | +1 (310) 516-8000 | |||
 | +1 (310) 516-7512 | |||
 | sales@spectrumchemical.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Cyanide/nitrile |
|---|---|
| Name | 4-Aminophthalonitrile |
| Synonyms | 3,4-Dicyanoaniline; 4-Amino-1,2-benzenedicarbonitrile |
| Molecular Structure | 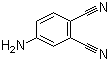 |
| Molecular Formula | C8H5N3 |
| Molecular Weight | 143.15 |
| CAS Registry Number | 56765-79-8 |
| EC Number | 260-370-6 |
| SMILES | C1=CC(=C(C=C1N)C#N)C#N |
| Melting point | 177-182 °C |
|---|---|
| Water solubility | Insoluble |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H315-H319-H331-H335 Details | ||||||||||||||||||||||||||||||||
| Safety Statements | P261-P262-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P319-P321-P330-P332+P317-P337+P317-P361+P364-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||
|
4-Aminophthalonitrile, a chemical compound also known as 4-APN, was first discovered through organic chemistry research. Its synthesis involves the reaction of phthalonitrile with ammonia or an amine derivative. This discovery can be traced back to the late 20th century, with its structure confirmed via spectroscopic analysis and chemical characterization. The unique combination of an amino group and phthalonitrile structure makes 4-Aminophthalonitrile a valuable compound for various applications across different industries. 4-Aminophthalonitrile serves as a key intermediate in the synthesis of vat dyes, which are widely used in the textile industry for dyeing cotton and other natural fibers. It contributes to the vibrant and colorfast hues of textile products. This compound is utilized as a building block in organic synthesis to produce various heterocyclic compounds and pharmaceutical intermediates. Its versatile reactivity enables the creation of complex molecules with diverse functionalities. 4-Aminophthalonitrile finds applications in materials science for the synthesis of functional materials, including polymers, resins, and coatings. It imparts specific properties such as thermal stability, electrical conductivity, and corrosion resistance to these materials. Used as a starting material in pharmaceutical synthesis, 4-Aminophthalonitrile contributes to the production of pharmaceuticals targeting various therapeutic areas, including cardiovascular diseases, cancer treatments, and antimicrobial agents. Researchers employ 4-Aminophthalonitrile in chemical research for studying reaction mechanisms and developing new synthetic routes. It serves as a valuable tool for exploring novel molecules and their potential applications in medicine, materials science, and other fields. References 2019. Generating the Blood Exposome Database Using a Comprehensive Text Mining and Database Fusion Approach. Environmental Health Perspectives, 127(9). DOI: 10.1289/ehp4713 2008. Synthesis of Phthalonitriles Using a Palladium Catalyst. Synlett, 2008(14). DOI: 10.1055/s-2008-1078269 2007. 4-acyloxy- and 4-acylaminophthalonitriles and phthalocyanines based thereon. Russian Journal of Organic Chemistry, 43(11). DOI: 10.1134/s1070428007110231 |
| Market Analysis Reports |