Online Database of Chemicals from Around the World
| AAT Bioquest, Inc. | USA | |||
|---|---|---|---|---|
 | www.aatbio.com | |||
 | +1 (408) 733-1055 | |||
 | +1 (408) 733-1304 | |||
 | jack@aatbio.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Scientia Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scientiapharm.net | |||
 | +86 (21) 5430-1573 6172-1573 400-036-1573 | |||
 | +86 (21) 5186-1981 | |||
 | sales@scientiapharm.com scientiapharm@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Hansa Fine Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.hfc-chemicals.com | |||
 | +49 (421) 427-682 | |||
 | +49 (421) 427-682 | |||
 | jbarten@hfc-chemicals.com | |||
| Chemical manufacturer | ||||
| Marker Gene Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.markergene.com | |||
 | +1 (888) 218-4062 (541) 342-3760 | |||
 | +1 (541) 342-1960 | |||
 | techservice@markergene.com | |||
| Chemical manufacturer since 1993 | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| SynChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.synchem.com | |||
 | +1 (847) 298-2436 ex 230 | |||
 | +1 (847) 298-2439 | |||
 | sales@synchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyran compound |
|---|---|
| Name | 7-Hydroxy-4-(trifluoromethyl)coumarin |
| Synonyms | 4-(Trifluoromethyl)umbelliferone |
| Molecular Structure | 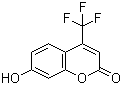 |
| Molecular Formula | C10H5F3O3 |
| Molecular Weight | 230.14 |
| CAS Registry Number | 575-03-1 |
| EC Number | 624-706-9 |
| SMILES | C1=CC2=C(C=C1O)OC(=O)C=C2C(F)(F)F |
| Density | 1.6±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 178 - 180 °C (Expl.) |
| Boiling point | 311.4±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 142.2±27.9 °C (Calc.)* |
| Index of refraction | 1.546 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H300-H315-H319-H335-H336 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
7-Hydroxy-4-(trifluoromethyl)coumarin is a fluorinated coumarin derivative with the molecular formula C10H5F3O3. It belongs to the class of 4-substituted coumarins, in which a trifluoromethyl group is attached at the 4-position and a hydroxy group at the 7-position. The compound was developed as part of efforts in the late 20th century to explore the chemical and photophysical properties of fluorinated coumarins. The presence of the trifluoromethyl group significantly increases the electron-withdrawing character at the 4-position, which affects the acidity of the 7-hydroxy group and modifies the overall reactivity and fluorescence properties of the molecule. The primary application of 7-hydroxy-4-(trifluoromethyl)coumarin is in fluorescence-based chemical analysis. The compound exhibits strong fluorescence, which can be modulated by chemical interactions, pH, or complexation with metal ions. This makes it a valuable probe for detecting and quantifying various analytes in biochemical and environmental studies. Its stability, fluorescence efficiency, and spectral properties are enhanced by the trifluoromethyl group, which also contributes to increased photostability and reduced susceptibility to photobleaching. In organic synthesis, 7-hydroxy-4-(trifluoromethyl)coumarin is used as a building block for the preparation of more complex fluorinated coumarin derivatives. The hydroxy group at the 7-position can participate in esterification, etherification, and condensation reactions, while the trifluoromethyl group can influence electronic properties, regioselectivity, and reactivity. This versatility allows chemists to design derivatives with tailored optical, chemical, or biological properties for research and industrial applications. In pharmaceutical and medicinal chemistry, coumarin derivatives such as 7-hydroxy-4-(trifluoromethyl)coumarin have been investigated for their potential as enzyme inhibitors, anticoagulants, and therapeutic agents. The trifluoromethyl group can enhance bioavailability, metabolic stability, and lipophilicity of derivatives, while the hydroxy functionality allows for further chemical modification. Although the parent compound is mainly used as an intermediate or fluorescent probe, its derivatives have potential applications in drug development and biological studies. In materials science, 7-hydroxy-4-(trifluoromethyl)coumarin has been used to produce fluorescent materials, dyes, and optical sensors. Incorporation of the trifluoromethyl group improves chemical and thermal stability, while the hydroxy group allows covalent attachment to polymer matrices or surfaces. These properties enable the design of functional materials for imaging, sensing, and photonic applications. The discovery and continued use of 7-hydroxy-4-(trifluoromethyl)coumarin highlight its importance as a versatile fluorinated coumarin derivative. Its combination of strong fluorescence, chemical stability, and functionalizable hydroxy group make it a valuable tool in analytical chemistry, organic synthesis, pharmaceutical research, and materials science. The compound exemplifies how selective fluorination can enhance the properties and utility of coumarin-based molecules in both scientific and industrial contexts. References 2019. BioTransformer: a comprehensive computational tool for small molecule metabolism prediction and metabolite identification. Journal of Cheminformatics, 11(1). DOI: 10.1186/s13321-018-0324-5 1998. Determination of the CYP2B6 Component of 7-Ethoxy-4-Trifluoromethylcoumarin O-Deethylation Activity in Human Liver Microsomes. Methods in Molecular Biology, 107. DOI: 10.1385/0-89603-519-0:117 1992. Synthesis of 4-trifluoromethyl-7-hydroxycoumarin derivatives. Structure of 3-cyano-4-trifluoromethyl-7-acetoxycoumarin. Bulletin of the Russian Academy of Sciences, Division of Chemical Science, 41(6). DOI: 10.1007/bf00866588 |
| Market Analysis Reports |