Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Changzhou A.H.(JOA) Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aihanchem.com | |||
 | +86 (519) 8560-9655 | |||
 | +86 (519) 8577-9936 | |||
 | sales@joachem.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shanghai ZaiQi Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemzq.com | |||
 | +86 (21) 5482-4098 | |||
 | +86 (21) 5482-4069 | |||
 | sales1@chemzq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| LONGRE Pharmaceuticals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.longrepharm.com | |||
 | +86 (512) 6959-2836 +86 13306132836 | |||
 | +86 (512) 6959-2835 | |||
 | sales@longrepharm.com yuguojun1013@163.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Activate Scientific GmbH | Germany | |||
|---|---|---|---|---|
 | www.activate-scientific.com | |||
 | +49 (8051) 965-1660 | |||
 | +49 (8051) 965-1661 | |||
 | rschmitt@activate-scientific.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 2-Chloro-4-nitroimidazole |
| Synonyms | 2-Chloro-4-nitro-1H-imidazole |
| Molecular Structure | 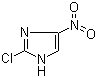 |
| Molecular Formula | C3H2ClN3O2 |
| Molecular Weight | 147.52 |
| CAS Registry Number | 57531-37-0 |
| EC Number | 611-554-3 |
| SMILES | C1=C(NC(=N1)Cl)[N+](=O)[O-] |
| Density | 1.7±0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 386.8±34.0 °C 760 mmHg (Calc.)* |
| Flash point | 187.7±25.7 °C (Calc.)* |
| Index of refraction | 1.631 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
2‑Chloro‑4‑nitro‑1H‑imidazole (C3H2ClN3O2) is a halogenated nitroimidazole compound with a chlorine atom at the 2‑position and a nitro group at the 4‑position of the imidazole ring. It is primarily used as a synthetic intermediate in organic chemistry, especially in the preparation of antitubercular drugs such as delamanid. Its chemical structure, combining the electron-withdrawing nitro group and the halogen substituent, enables selective reactions at different positions of the heterocyclic ring, making it versatile for downstream chemical transformations. The nitro group at the 4‑position can be reduced to an amino group, which allows the formation of derivatives essential for the synthesis of complex pharmaceuticals. The chlorine atom at the 2‑position facilitates regioselective substitution reactions, including palladium-catalyzed cross-couplings, which are widely used in medicinal chemistry for constructing more elaborate molecular scaffolds. 2‑Chloro‑4‑nitroimidazole can also undergo N‑alkylation, providing further functionalization possibilities for synthetic applications. The compound has a nearly planar molecular structure, and in the solid state, molecules are arranged through intermolecular hydrogen bonding, forming two-dimensional networks. This crystalline arrangement is important for its stability and ease of handling during synthesis. As an intermediate, 2‑Chloro‑4‑nitroimidazole is critical in the pharmaceutical industry for producing nitroimidazole-based anti-tuberculosis agents. Its structural features are integral to the biological activity of these drugs, particularly under hypoxic conditions where tuberculosis bacteria are less susceptible to standard treatments. Handling 2‑Chloro‑4‑nitroimidazole requires appropriate safety measures. It is potentially irritant and should be stored in a cool, dry place with adequate protective equipment used during manipulation. Its stability and reactivity make it a practical and widely used chemical intermediate in both academic and industrial laboratories. References Fun HK, Ng SW, Wong WT (2010) Crystal structure of 2‑chloro‑4‑nitro‑1H‑imidazole. Acta Crystallographica Section E 66: o1828–o1829 DOI: 10.1107/S1600536810029913 Showalter HD, Awasthi D, Singh R, Et al. (2020) Recent progress in the discovery and development of 2‑halo‑4‑nitroimidazoles as anti‑tuberculosis agents. Molecules 25(18): 4137 DOI: 10.3390/molecules25184137 Thompson AM, O’Connor PD, Blaser A, et al. (2017) Structural‑activity relationships of antitubercular nitroimidazoles: insights from authentic 3‑nitro isomer of pretomanid. ACS Medicinal Chemistry Letters 8(8): 854–858 DOI: 10.1021/acsmedchemlett.7b00356 |
| Market Analysis Reports |