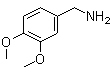
Veratrylamine is an organic compound classified as a phenethylamine derivative, characterized by a 3,4-dimethoxyphenyl group attached to an ethylamine chain. It is chemically related to naturally occurring alkaloids found in certain plant species, especially those of the Veratrum genus, from which its name is derived. The compound is notable for its role as a bioactive intermediate in the synthesis of various pharmacologically important molecules.
The initial isolation and identification of veratrylamine trace back to studies on Veratrum alkaloids, where it was found as a key structural component or biosynthetic precursor. These alkaloids have been traditionally used in medicine and ethnobotany for their physiological effects. Veratrylamine itself was studied for its potential pharmacological activity, including interactions with adrenergic receptors and effects on the central nervous system.
In modern chemical research, veratrylamine is mainly utilized as a synthetic building block. Its functional groups and aromatic substitution pattern make it a versatile intermediate for the preparation of complex molecules, including pharmaceutical agents such as vasodilators, antihypertensives, and neuroactive compounds. The dimethoxyphenyl moiety contributes to specific binding interactions and enhances lipophilicity, which is important in drug design.
Veratrylamine can be synthesized through various routes, including the reductive amination of 3,4-dimethoxyphenylacetone or via demethylation and amination processes starting from related natural or synthetic precursors. Its chemical reactivity allows further modification at the amine group or aromatic positions, facilitating the creation of diverse derivatives for structure-activity relationship studies.
Pharmacologically, veratrylamine and its derivatives have been examined for their potential to influence neurotransmitter systems, particularly by acting as precursors or analogs to compounds affecting dopamine and norepinephrine pathways. This has relevance in research on neurological disorders and cardiovascular regulation.
Due to its bioactive properties, veratrylamine is handled under controlled laboratory conditions with appropriate safety measures. It is generally regarded as a valuable compound in medicinal chemistry research rather than as a commercial drug itself.
In summary, veratrylamine is a phenethylamine derivative originally identified as part of Veratrum alkaloids. It serves primarily as a synthetic intermediate in pharmaceutical chemistry and has been investigated for its biological activities related to the nervous and cardiovascular systems. Its well-established chemical and pharmacological properties make it an important compound in both natural product chemistry and drug development.
References
1997. 3,4-Dimethoxybenzylamine as a sensitive pre-column fluorescence derivatization reagent for the determination of serotonin in human platelet-poor plasma. Journal of Chromatography B: Biomedical Sciences and Applications, 692(1).
DOI: 10.1016/s0378-4347(96)00481-1
2006. Reactions of Cyanomethanesulfonamides with Aldehydes and Synthesis of 2-Benzyl-2,3-dihydrobenzopyrano[3,2-e][1,2,4]thiadiazine 1,1-Dioxides. Monatshefte für Chemie / Chemical Monthly, 137(10).
DOI: 10.1007/s00706-006-0541-x
2018. Synthesis and Neuroprotective Activity of Neolamellarin A Analogues. Journal of Ocean University of China, 17(4).
DOI: 10.1007/s11802-018-3530-x
|







































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details