Online Database of Chemicals from Around the World
| Shanghai Send Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shsendpharma.com | |||
 | +86 (0)21) 5808-8081 | |||
 | sale@shsendpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | 3-(1H-indol-3-yl)propanamide |
| Molecular Structure | 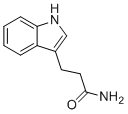 |
| Molecular Formula | C11H12N2O |
| Molecular Weight | 188.23 |
| CAS Registry Number | 5814-93-7 |
| EC Number | 855-919-7 |
| SMILES | C1=CC=C2C(=C1)C(=CN2)CCC(=O)N |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.662, Calc.* |
| Boiling Point | 487.6±28.0 °C (760 mmHg), Calc.* |
| Flash Point | 248.7±24.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-(1H-indol-3-yl)propanamide is an organic compound that belongs to the class of indole derivatives, which have a wide range of applications in chemical research and pharmaceutical development. Structurally, this compound consists of an indole ring attached to a three-carbon chain with an amide functional group. The indole core is a highly significant structure in medicinal chemistry due to its presence in many biologically active molecules, including neurotransmitters like serotonin. The discovery of 3-(1H-indol-3-yl)propanamide is tied to the broader exploration of indole derivatives, which have been studied extensively for their biological properties. Researchers have been particularly interested in modifications of the indole structure to develop new therapeutic agents for various diseases, including cancer, inflammation, and neurological disorders. The addition of an amide group to the indole ring at the 3-position, as seen in 3-(1H-indol-3-yl)propanamide, alters the compound's chemical reactivity and potential biological activity, making it an attractive candidate for drug discovery. In terms of application, 3-(1H-indol-3-yl)propanamide has been investigated as a precursor in the synthesis of more complex bioactive molecules. It serves as a building block for the development of compounds with potential pharmacological properties. Indole derivatives with similar structures have shown promise in areas such as anti-inflammatory, anti-cancer, and anti-microbial research. This particular compound may also be used in research focused on enzyme inhibition or receptor modulation, given the bioactivity typically associated with indole-based molecules. Additionally, the amide group in 3-(1H-indol-3-yl)propanamide can engage in hydrogen bonding, which can be leveraged in drug design to improve the compound’s interaction with biological targets. This feature makes it a versatile intermediate for the development of drug candidates, where the optimization of solubility, permeability, and target affinity is critical for therapeutic efficacy. As the pharmaceutical industry continues to explore the potential of indole derivatives, compounds like 3-(1H-indol-3-yl)propanamide play a crucial role in the synthesis of novel therapeutic agents. The ability to modify this structure further enhances its utility in drug discovery programs focused on addressing unmet medical needs. References 2015. Characterization of an Antibacterial Compound, 2-Hydroxyl Indole-3-Propanamide, Produced by Lactic Acid Bacteria Isolated from Fermented Batter. Applied Biochemistry and Biotechnology, 177(1). DOI: 10.1007/s12010-015-1733-9 2009. A Novel Indole-3-propanamide Exerts Its Immunosuppressive Activity by Inhibiting JAK3 in T Cells. The Journal of Pharmacology and Experimental Therapeutics, 331(2). DOI: 10.1124/jpet.109.155986 2001. Synthesis and analytical evaluation by voltammetric studies of some new indole-3-propionamide derivatives. Farmaco (Societa chimica italiana : 1989), 56(11). DOI: 10.1016/s0014-827x(01)01150-8 |
| Market Analysis Reports |