Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Inorganic chemical industry >> Inorganic salt >> Carbides and carbonates |
|---|---|
| Name | Lanthanum Carbonate |
| Molecular Structure | 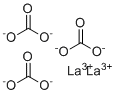 |
| Molecular Formula | C3La2O9 |
| Molecular Weight | 457.84 |
| CAS Registry Number | 587-26-8 |
| EC Number | 209-599-5 |
| SMILES | C(=O)([O-])[O-].C(=O)([O-])[O-].C(=O)([O-])[O-].[La+3].[La+3] |
| Density | 2.6-2.7 g/cm3 |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
|
Lanthanum carbonate (La2(CO3)3) is a pharmaceutical compound used primarily to treat hyperphosphatemia in patients with chronic kidney disease (CKD). Lanthanum carbonate was discovered during the ongoing search for an effective phosphate binder. Hyperphosphatemia, characterized by too much phosphate in the blood, is a common problem in CKD patients due to impaired kidney function. This condition can lead to cardiovascular and bone disease. Traditional treatments have limitations, prompting researchers to explore alternative compounds. Before the advent of lanthanum carbonate, aluminum-based phosphate binders were widely used but had toxicity issues, including aluminum accumulation and bone disease. Another option was calcium-based binders, but there was a risk of hypercalcemia (high calcium levels) and vascular calcification. Therefore, the discovery of lanthanum carbonate provided a safer and more effective alternative. Lanthanum carbonate has the chemical formula La2(CO3)3 and consists of lanthanum ions coordinated to carbonate ions. Its chemical properties allow it to effectively bind dietary phosphate in the gastrointestinal tract, preventing it from being absorbed into the blood. Lanthanum is a rare earth element whose medical applications exploit its unique ability to form stable, insoluble complexes with phosphate. Lanthanum carbonate works by binding to phosphate in the digestive system to form insoluble lanthanum phosphate complexes. These complexes are excreted in the feces, thereby reducing the amount of phosphate absorbed into the blood. This mechanism helps to reduce serum phosphate levels in patients with CKD, thereby mitigating the risks associated with hyperphosphatemia. Lanthanum carbonate is used as a phosphate binder in patients with CKD, especially those on dialysis. It helps to control serum phosphate levels, thereby reducing the risk of secondary hyperparathyroidism, cardiovascular disease, and bone disease. As a non-calcium-based binder, lanthanum carbonate is beneficial for patients who need to avoid calcium overload. This makes it suitable for individuals with hypercalcemia or risk of vascular calcification. Lanthanum carbonate supplements dietary phosphate restrictions, allowing for more flexibility in managing phosphate intake while still effectively controlling blood phosphate levels. Lanthanum carbonate is generally well tolerated with a favorable safety profile. Common side effects include gastrointestinal symptoms such as nausea, vomiting, and diarrhea. Unlike aluminum-based adhesives, it does not cause metal accumulation. However, long-term monitoring is essential to ensure its continued effectiveness and safety. References 2024. Effects of the combined use of lanthanum carbonate and activated carbon capping materials on phosphorus and dissolved organic matter in lake sediments. Environmental Research. DOI: 10.1016/j.envres.2024.120291 2024. Simultaneous immobilization of sediment internal phosphorus, arsenic and tungsten by lanthanum carbonate capping. Environmental Research. DOI: 10.1016/j.envres.2023.117817 2024. Effect of Lanthanum Carbonate on Serum Phosphate, Oxidative Stress, and Vascular Dysfunction in CKD: A Mechanistic Randomized Controlled Trial. Kidney360. DOI: 10.34067/kid.0000000000000465 |
| Market Analysis Reports |