Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Xuzhou Ruisai Technology Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.risachem.com | |||
 | +86 (526) 8776-8018 | |||
 | +86 (526) 8776-8008 | |||
 | sales@risachem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Reddy N Reddy Pharmaceuticals | India | |||
|---|---|---|---|---|
 | www.reddynreddypharma.com | |||
 | +91 (40) 2307-5686 +91 98480-96759 +91 98481-96759 | |||
 | info@reddynreddypharma.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Aromsyn Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aromsyn.com | |||
 | +86 (571) 8558-5865 +86 13336024896 | |||
 | +86 (571) 8558-5865 | |||
 | service@aromsyn.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Wonderchem Co., Limited | Hong Kong | |||
|---|---|---|---|---|
 | www.wonder-chem.com | |||
 | +852 8192-5218 | |||
 | sales@wonder-chem.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Raymon Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raymonbio.com | |||
 | +86 (25) 5218-1235 | |||
 | sales@raymonbio.com | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Syntech Labs, Inc. | USA | |||
|---|---|---|---|---|
 | www.syntechlabs.com | |||
 | +1 (732) 545-8380 | |||
 | +1 (732) 545-6881 | |||
 | info@syntechlabs.com | |||
| Chemical manufacturer | ||||
| Aaron Chemistry GmbH | Germany | |||
|---|---|---|---|---|
 | www.aaron-chemistry.de | |||
 | +49 (8823) 917-521 | |||
 | +49 (8823) 917-523 | |||
 | sales@aaron-chemistry.de | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Organica Feinchemie GmbH | Germany | |||
|---|---|---|---|---|
 | www.organica.de | |||
 | +49 (3494) 63-6215 | |||
 | +49 (3494) 63-6165 | |||
 | dmi@weylchem-organica.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic aldehyde (containing acetal, hemiacetal) |
|---|---|
| Name | 3-Methoxybenzaldehyde |
| Synonyms | m-Anisaldehyde |
| Molecular Structure | 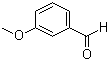 |
| Molecular Formula | C8H8O2 |
| Molecular Weight | 136.15 |
| CAS Registry Number | 591-31-1 |
| EC Number | 209-712-8 |
| SMILES | COC1=CC=CC(=C1)C=O |
| Density | 1.119 |
|---|---|
| Boiling point | 143 °C (50 mmHg) |
| Refractive index | 1.55-1.554 |
| Flash point | 110 °C |
| Water solubility | insoluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
3-Methoxybenzaldehyde, a benzaldehyde derivative with a methoxy group attached to the aromatic ring, is a significant compound in organic chemistry. Its unique structure, characterized by a methoxy group (-OCH₃) attached to the 3-position of the benzaldehyde ring, lends itself to various chemical reactions and applications across multiple fields. The discovery of 3-methoxybenzaldehyde dates back to the early 20th century when researchers explored the reactivity and applications of substituted benzaldehydes. The methoxy group, known for its electron-donating properties, significantly influences the chemical behavior of the benzaldehyde ring. This electron-donating effect enhances the reactivity of the aldehyde group, making 3-methoxybenzaldehyde a valuable intermediate in organic synthesis. In synthetic organic chemistry, 3-methoxybenzaldehyde serves as a precursor for various chemical transformations. Its aldehyde group is highly reactive, participating in condensation reactions such as the formation of Schiff bases and imines. The methoxy group, due to its electron-donating nature, facilitates these reactions by stabilizing the intermediates formed during the process. This makes 3-methoxybenzaldehyde an essential building block in the synthesis of complex organic molecules. One of the notable applications of 3-methoxybenzaldehyde is in the production of pharmaceuticals. It is used in the synthesis of various bioactive compounds, including anti-inflammatory agents, anti-cancer drugs, and antidepressants. The methoxy group’s influence on the electron density of the benzaldehyde ring can modify the pharmacological properties of the final compounds, making 3-methoxybenzaldehyde a crucial intermediate in drug development. In addition to its pharmaceutical applications, 3-methoxybenzaldehyde is employed in the synthesis of flavor and fragrance compounds. Its aromatic profile, characterized by a sweet and slightly floral odor, makes it suitable for use in perfumery and flavoring industries. The compound’s ability to form various derivatives with distinct sensory properties expands its utility in creating diverse fragrance and flavor profiles. 3-Methoxybenzaldehyde also finds applications in materials science. It is used as a starting material in the synthesis of organic materials with specific properties, such as dyes and polymers. The compound’s reactivity allows for the introduction of the methoxy group into various polymer backbones, influencing the material's properties such as solubility, color, and stability. Overall, 3-methoxybenzaldehyde is a versatile compound with a broad range of applications in organic synthesis, pharmaceuticals, flavor and fragrance industries, and materials science. Its unique structure and reactivity make it a valuable intermediate in the creation of diverse chemical products and materials. References 1948. Structure Determination and Synthesis of a Plant Growth Inhibitor, 3-Acetyl-6-methoxybenzaldehyde, Found in the Leaves of Encelia Farinosa. Journal of the American Chemical Society, 70(3). DOI: 10.1021/ja01183a114 2024. Recent advances in the synthesis of dihydropyrimidinones via biginelli reaction. Journal of the Iranian Chemical Society, 21(10). DOI: 10.1007/s13738-024-03100-z 2024. Synthesis of Hantzsch poly-substituted pyridines containing adamantyl moiety. Monatshefte für Chemie - Chemical Monthly, 155(2). DOI: 10.1007/s00706-023-03164-2 |
| Market Analysis Reports |