Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Molecraft LifeSciences Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.molecraft.com | |||
 | +91 (91) 6018-8881 | |||
 | +91 (40) 2359-0011 | |||
 | info@molecraft.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| VanDeMark Chemical Inc. | USA | |||
|---|---|---|---|---|
 | www.vandemark.com | |||
 | +1 (716) 433-6764 | |||
 | +1 (716) 433-2850 | |||
 | sales@vandemark.com | |||
| Chemical manufacturer since 1951 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cn Chemunion Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cnchemunion.com | |||
 | +86 17366116869 | |||
 | david@cnchemunion.com | |||
 | QQ Chat | |||
 | WeChat: +86 15250069576 | |||
 | WhatsApp:+86 17366116869 | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Changzhou Suntton Co., Ltd. | USA | |||
|---|---|---|---|---|
 | www.suntton.com | |||
 | +1 (914) 965-2077 +86 (519) 8513-6089 | |||
 | +1 (914) 375-2093 / +86 (519) 8513-6133 | |||
 | luke.verdet@suntton.com lily.chen@suntton.com | |||
| Chemical manufacturer | ||||
| Sarchem Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.sarchemlabs.com | |||
 | +1 (732) 708-1777 | |||
 | +1 (732) 708-1265 | |||
 | sarchem@aol.com | |||
| Chemical manufacturer since 1984 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Butyl ester compound |
|---|---|
| Name | Butyl chloroformate |
| Synonyms | n-Butyl Chloroformate |
| Molecular Structure | 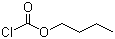 |
| Molecular Formula | C5H9ClO2 |
| Molecular Weight | 136.58 |
| CAS Registry Number | 592-34-7 |
| EC Number | 209-750-5 |
| SMILES | CCCCOC(=O)Cl |
| Density | 1.074 |
|---|---|
| Melting point | -70 °C |
| Boiling point | 142 °C |
| Refractive index | 1.411-1.413 |
| Flash point | 39 °C |
| Water solubility | DECOMPOSES |
| Hazard Symbols |     GHS02;GHS06;GHS05;GHS07 Danger Details GHS02;GHS06;GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H331-H314 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P284-P301+P317-P301+P330+P331-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P320-P321-P330-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 2743 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Butyl chloroformate, with the chemical formula C5H9ClO2, is an organochlorine compound known for its role as a reagent in organic synthesis. This compound features a butyl group attached to a chloroformate moiety, which makes it a versatile tool in the preparation of various chemical derivatives. Its discovery and development have significantly impacted its applications in organic chemistry, particularly in the synthesis of esters, carbonates, and other functional groups. The discovery of butyl chloroformate is rooted in the exploration of chloroformates and their derivatives, which have been used as reactive intermediates in organic synthesis. Chloroformates, characterized by their ability to react with alcohols and amines, play a crucial role in introducing ester and carbonate functionalities into molecules. The synthesis of butyl chloroformate typically involves the reaction of butanol with phosgene, resulting in the formation of the chloroformate ester. In organic synthesis, butyl chloroformate serves as a valuable reagent for protecting alcohols and amines during multi-step syntheses. When reacting with alcohols, it forms butyl esters, which are stable and can be selectively removed after the desired transformations are complete. Similarly, butyl chloroformate reacts with amines to produce butyl carbamates, which are used to protect amino groups during complex chemical processes. This protective strategy is essential for ensuring selectivity and preventing undesired side reactions in multi-step syntheses. Butyl chloroformate also plays a role in the synthesis of pharmaceuticals and fine chemicals. Its ability to introduce carbonyl groups into molecules makes it valuable in the preparation of carbonates and carbamates, which are important functional groups in drug development and other chemical applications. The compound's versatility extends to the synthesis of polymer precursors and specialty chemicals, demonstrating its utility in various industrial applications. In summary, butyl chloroformate is a significant reagent in organic chemistry, providing valuable applications in the protection of functional groups and the synthesis of complex molecules. Its discovery and development have enhanced the capabilities of organic synthesis, making it an essential tool in the preparation of pharmaceuticals, polymers, and other fine chemicals. References 2022. Novel Self-Assembled Micelles With Increased Tumor Penetration and Anti-Tumor Efficiency Against Breast Cancer. Pharmaceutical Research. DOI: 10.1007/s11095-022-03338-3 2020. Development and validation of a gas chromatography-mass spectrometry method to analyze octanoate enrichments at low concentrations in human plasma. Analytical and Bioanalytical Chemistry. DOI: 10.1007/s00216-020-02801-7 2016. Application of Solid-Phase Microextraction Combined with Derivatization for Polar Compound Sampling in Environmental Analysis. Solid Phase Microextraction. DOI: 10.1007/978-3-662-53598-1_7 |
| Market Analysis Reports |