Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hebei Jianxin Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jianxinchemical.com | |||
 | +86 (317) 356-7500 356-2699 | |||
 | +86 (317) 350-1550 / 359-5999 | |||
 | sales@jianxinchemical.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Witofly Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.witofly.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales@witofly.com | |||
 | QQ Chat | |||
| Chemical distributor since 2016 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Chengdu Yuanda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ydachem.com | |||
 | +86 (28) 8535-6300 | |||
 | +86 (28) 8535-6300 | |||
 | sales@ydachem.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Wuhan Comings Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | comings.en.made-in-china.com | |||
 | +86 13659820406 | |||
 | info@comingschemi.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Wuxi Cidic Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cidic.com.cn | |||
 | +86 (510) 8230-3030 | |||
 | +86 (510) 8230-3031 | |||
 | baoming@cidic.com.cn | |||
| Chemical distributor | ||||
| Classification | Chemical reagent >> Organic reagent >> Sulfone, sulfoxide compound |
|---|---|
| Name | 3,3'-Sulfonyldianiline |
| Synonyms | 3-Aminophenyl sulfone; 3,3'-Diamino diphenylsulfone; Bis(3-aminophenyl) sulfone |
| Molecular Structure | 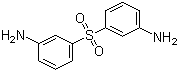 |
| Molecular Formula | C12H12N2O2S |
| Molecular Weight | 248.30 |
| CAS Registry Number | 599-61-1 |
| EC Number | 209-967-5 |
| SMILES | C1=CC(=CC(=C1)S(=O)(=O)C2=CC=CC(=C2)N)N |
| Water solubility | slightly soluble |
|---|---|
| Density | 1.4$+/-$0.1 g/cm3 Calc.* |
| Melting point | 170 - 173 $degree$C (Expl.) |
| Boiling point | 542.8$+/-$35.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 282.1$+/-$25.9 $degree$C (Calc.)* |
| Index of refraction | 1.662 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H371-H373-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P308+P316-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
3,3'-Sulfonyldianiline is an aromatic diamine compound in which two amino groups are attached to benzene rings that are linked via a central sulfone (–SO2–) group at the 3-position of each ring. This structure combines the reactivity of the amino groups with the electron-withdrawing and thermally stable properties of the sulfone moiety, making it a valuable intermediate in polymer chemistry, organic synthesis, and high-performance material development. The sulfone linkage introduces rigidity and thermal stability, while the amino groups allow for further functionalization. The molecular structure consists of two benzene rings connected through a sulfone functional group, with each benzene ring substituted with a primary amino group at the meta position relative to the sulfone. The amino groups provide nucleophilic sites that can participate in reactions such as acylation, sulfonylation, and polycondensation, while the sulfone bridge enhances electron deficiency on the aromatic rings and increases thermal resistance. This combination of reactive and stabilizing features makes the molecule particularly suitable as a monomer for advanced polymer synthesis. Synthesis of 3,3'-sulfonyldianiline typically involves the sulfonation of 3-aminobenzenes followed by oxidative coupling to form the sulfone-linked diamine. Alternative approaches may include direct oxidation of bis(3-aminophenyl)sulfide to the corresponding sulfone. Careful control of reaction conditions, such as temperature, oxidizing agent, and solvent, is required to ensure selective formation of the sulfone without overoxidation or side reactions. The product is obtained as a solid with high purity and stability, suitable for downstream applications. One of the primary applications of 3,3'-sulfonyldianiline is in the production of high-performance polymers, including polyamides, polyimides, and epoxy resins. The diamine functionality allows polymer chain extension and crosslinking, while the sulfone group imparts thermal stability, mechanical strength, and chemical resistance. Polymers derived from this diamine are used in electronics, coatings, adhesives, and composites where high thermal and chemical stability is essential. In addition to polymer applications, 3,3'-sulfonyldianiline serves as a building block in organic synthesis for preparing functional aromatic compounds, heterocycles, and dye intermediates. Its combination of nucleophilic amino groups and electron-withdrawing sulfone linkage allows for selective reactions and modification to produce molecules with tailored chemical and physical properties. Overall, 3,3'-sulfonyldianiline is a multifunctional aromatic diamine with significant utility in high-performance polymer synthesis, organic chemistry, and materials science. Its combination of nucleophilic amino groups, rigid sulfone linkage, and thermal stability provides both reactivity and durability, making it an important intermediate for the preparation of advanced materials and functional chemical compounds. References 2025. A novel naphthyl amine curing agent and its effect on curing performance of AF-type phthalonitrile resin. Iranian Polymer Journal. DOI: 10.1007/s13726-025-01524-9 2025. Cellulose nanocrystals effect to improve mechanical properties of epoxy-based glass fiber-reinforced composites. Iranian Polymer Journal. DOI: 10.1007/s13726-025-01468-0 2024. Effects of Ether Linkage and Benzene Ring Substitution Structures on Thermal and Optical Properties of 6FDA/3DDS-Based Transparent Polyimide Films. Fibers and Polymers. DOI: 10.1007/s12221-024-00793-0 |
| Market Analysis Reports |