Nickel(II) acetate tetrahydrate, with the chemical formula Ni(C2H3O2)2·4H2O, is an inorganic compound characterized by its vibrant green color and crystalline structure. This compound is a hydrated form of nickel acetate, and it has garnered interest due to its diverse applications in various fields, including chemistry, materials science, and biochemistry. Nickel(II) acetate was first synthesized in the early 19th century by the reaction of acetic acid with nickel oxide or nickel carbonate, establishing a basis for its use in laboratories and industries.
One of the primary applications of nickel(II) acetate tetrahydrate is in the preparation of nickel catalysts for organic synthesis. It is utilized in various catalytic reactions, including hydrogenation and cross-coupling reactions, due to its ability to provide nickel ions that facilitate these processes. Nickel catalysts are increasingly being favored for their cost-effectiveness and lower toxicity compared to their precious metal counterparts, making nickel(II) acetate a valuable component in sustainable chemistry practices.
In materials science, nickel(II) acetate tetrahydrate plays a crucial role in the production of nickel-containing materials, such as nickel oxide and nickel hydroxide. These materials are essential for various applications, including battery technology, where nickel oxides are used as cathode materials in rechargeable batteries. The ability to control the particle size and morphology of nickel oxide through the use of nickel(II) acetate tetrahydrate during synthesis allows for the optimization of the performance of these materials.
Another significant application of nickel(II) acetate tetrahydrate lies in the field of electroplating. It serves as a nickel source in electroplating baths, where nickel ions are deposited onto substrates to create protective or decorative coatings. The use of nickel coatings is widespread in industries such as automotive, electronics, and jewelry, where enhanced corrosion resistance and aesthetic appeal are crucial.
In biochemistry, nickel(II) acetate tetrahydrate is of interest due to its role in enzymatic processes. Nickel is an essential trace element in certain organisms, and nickel(II) acetate can be used to study nickel-dependent enzymes. These enzymes play vital roles in various biological pathways, including urease activity, and understanding their mechanisms can provide insights into metabolic processes and potential applications in biotechnology.
Moreover, nickel(II) acetate tetrahydrate has applications in the production of pigments and dyes, where it is utilized as a precursor in the synthesis of nickel-based colorants. These pigments are employed in coatings, plastics, and ceramics, contributing to the production of vibrant and stable colors.
In summary, nickel(II) acetate tetrahydrate is a versatile compound with significant applications across multiple fields, including catalysis, materials science, electroplating, biochemistry, and pigment production. Its discovery has facilitated advancements in sustainable chemistry and materials development, showcasing the importance of nickel compounds in both industrial and scientific domains.
References
2013. The Merck Index, An Encyclopedia of Chemicals, Drugs, and Biologicals, 15th Ed. Cambridge, UK: The Royal Society of Chemistry. URL: https://www.rsc.org/merck-index
1980. Gekkan Yakuji. *Pharmaceuticals Monthly*, 22(455). No DOI or URL available.
1980. Research Communications in Chemical Pathology and Pharmacology, 30(133). URL: https://pubmed.ncbi.nlm.nih.gov/7433764
|












































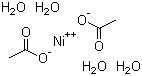

 GHS07;GHS08 Danger Details
GHS07;GHS08 Danger Details