Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai Oripharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oripharm.com.cn | |||
 | +86 (21) 6439-6936 | |||
 | +86 (21) 6481-4386 | |||
 | info@oripharm.com.cn sales@oripharm.com.cn | |||
| Chemical distributor since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Manus Aktteva | India | |||
|---|---|---|---|---|
 | www.manusaktteva.in | |||
 | +91 (79) 6512-3395 | |||
 | +91 (79) 2646-3395 | |||
 | products@manusakttevabiopharma.in | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhu Huaren Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huarensh.com | |||
 | +86 (553) 584-2013 | |||
 | +86 (553) 584-3138 | |||
 | wuhuhuaren@163.com | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Micro-Mega Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.micro-megaindustry.com | |||
 | +86 (21) 5715-3848 3462-8682 | |||
 | +86 (21) 5715-3848 | |||
 | weij@micro-megaindustry.com lewisliu2010@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Chemgenes India Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.chemgenesindia.com | |||
 | +91 (868) 742-1036 | |||
 | info@chemgenesindia.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Xinxiang Aurora Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aurora-biotech.com | |||
 | +86 (373) 308-8722 +86 18637352520 | |||
 | +86 (373) 308-8722 | |||
 | info@aurora-biotech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shenzhen Nexconn Pharmatechs Ltd. | China | |||
|---|---|---|---|---|
 | www.nexconn.com | |||
 | +86 19068605196 | |||
 | jason.deng@nexconn.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Berry & Associates, Inc. | USA | |||
|---|---|---|---|---|
 | www.berryassoc.com | |||
 | +1 (734) 426-3787 | |||
 | +1 (734) 426-9077 | |||
 | orders@berryassoc.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Nucleoside drugs >> Deoxynucleotides and their analogues |
|---|---|
| Name | 5-Iodo-2'-deoxycytidine |
| Synonyms | 4-Amino-1-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodopyrimidin-2-one; 5-IdC |
| Molecular Structure | 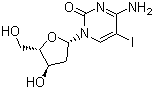 |
| Molecular Formula | C9H12IN3O4 |
| Molecular Weight | 353.11 |
| CAS Registry Number | 611-53-0 |
| EC Number | 210-269-8 |
| SMILES | C1[C@@H]([C@H](O[C@H]1N2C=C(C(=NC2=O)N)I)CO)O |
| Density | 2.4±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.815, Calc.* |
| Boiling Point | 522.1±60.0 °C (760 mmHg), Calc.* |
| Flash Point | 269.6±32.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
5-Iodo-2'-deoxycytidine is a synthetic nucleoside analog primarily used in molecular biology and medicinal chemistry. It is a derivative of 2'-deoxycytidine, where the hydrogen atom at the 5-position of the pyrimidine ring is replaced with an iodine atom. This modification allows it to interfere with DNA synthesis and replication processes, making it useful in various applications. The synthesis of 5-iodo-2'-deoxycytidine was first reported in the early 1970s. As a nucleoside analog, it was designed to explore the potential of halogenated compounds in inhibiting viral replication and cell proliferation, particularly in cancer therapy. It is closely related to other halogenated nucleosides that have shown potential in chemotherapy and antiviral treatments. 5-Iodo-2'-deoxycytidine has demonstrated antiviral properties, particularly against DNA viruses. The iodine substitution at the 5-position of the pyrimidine ring influences the incorporation of the nucleoside into viral DNA, inhibiting viral replication. It has been studied for its effects on viruses such as herpes simplex virus (HSV) and human cytomegalovirus (HCMV). As a nucleoside analog, 5-iodo-2'-deoxycytidine is also studied for its potential in cancer treatment. It can be incorporated into the DNA of rapidly dividing cancer cells, disrupting DNA replication and inducing cytotoxicity. By interfering with the DNA replication machinery, it inhibits cell proliferation and promotes apoptosis, making it a candidate for chemotherapy, particularly in the treatment of solid tumors. 5-Iodo-2'-deoxycytidine is a substrate for the enzyme deoxycitidine kinase, which phosphorylates it to its active triphosphate form. This active form is then incorporated into the DNA during replication. The halogenated base alters the structure of the DNA and inhibits the function of DNA polymerases, leading to DNA damage and cellular death. This mechanism is particularly effective in rapidly dividing cells, such as cancer cells or virus-infected cells. Beyond its direct therapeutic potential, 5-iodo-2'-deoxycytidine is used in research, particularly in studies of nucleic acid metabolism and DNA replication. Its incorporation into DNA and the subsequent effects on cellular processes make it a valuable tool for studying DNA replication, repair, and cell cycle regulation. In conclusion, 5-iodo-2'-deoxycytidine is a halogenated nucleoside analog with applications in antiviral therapy, cancer treatment, and molecular research. Its ability to inhibit DNA replication makes it an important compound for both therapeutic and investigative purposes, particularly in oncology and virology. References 1961. Preparation of 5-iodo-2'-deoxycytidine. Biochemical Pharmacology, 8(2). DOI: 10.1016/0006-2952(61)90109-5 1998. Efficacy of select antivirals against Cryptosporidium parvum in vitro. FEMS Microbiology Letters, 168(1). DOI: 10.1016/s0378-1097(98)00419-4 2018. New Synthetic Route to CY5-Labeled 2'-Deoxycytidine-5'-Triphosphates Using Sonogashira Reaction. Russian Journal of Bioorganic Chemistry, 44(2). DOI: 10.1134/s1068162018020103 |
| Market Analysis Reports |