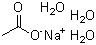Sodium acetate trihydrate (NaC2H3O2·3H2O) has been known since the 18th century. The compound is produced by neutralizing acetic acid with sodium carbonate or sodium hydroxide. Sodium acetate crystallizes with three water molecules to form a trihydrate. This form is a colorless or white crystalline solid that releases water when heated.
In the food industry, sodium acetate trihydrate is used as a preservative and flavoring agent. It helps control acidity and acts as a buffer to maintain the desired pH in products such as sauces, condiments, and baked goods. It extends shelf life and stability while imparting a mild salty flavor that improves mouthfeel. This makes it an indispensable ingredient in processed foods where maintaining quality and safety is critical.
Sodium acetate trihydrate also plays an important role in the medical field. It is used as an electrolyte supplement in intravenous (IV) fluids to help patients maintain proper hydration and electrolyte balance. Its buffering capacity is essential for regulating pH in various pharmaceutical formulations, improving drug stability and efficacy.
In addition to food and pharmaceuticals, sodium acetate trihydrate is used in many industrial processes. It acts as a chemical buffer in dyed textiles, aiding in the absorption of dyes by stabilizing pH. In the paper industry, it is used to neutralize acids, preventing the degradation of paper quality. Additionally, it softens water and aids in the removal of impurities in water treatment, contributing to environmental sustainability.
One notable application of sodium acetate trihydrate is in reusable heating pads. These pads use a supersaturated solution of sodium acetate trihydrate and, when triggered, crystallize exothermically, releasing heat. This property is often demonstrated in "hot ice" experiments, demonstrating the compound's ability to provide a portable heat source. This is particularly useful for thermal applications in therapeutic settings and in environments where electrical heating is impractical.
In household cleaning products, sodium acetate trihydrate acts as a buffer, increasing the effectiveness of detergents and cleaning solutions by maintaining an optimal pH. Its mild corrosive properties help remove mineral deposits and stains, making it a useful ingredient in a variety of cleaners.
Sodium acetate trihydrate is safe when used appropriately. However, as with all chemicals, it should be handled with care. In industrial settings, proper storage and handling procedures are essential to prevent accidental exposure or misuse. In consumer products, formulations are required to ensure safe exposure levels.
Sodium acetate trihydrate is biodegradable and has a relatively low environmental impact. It is produced from acetic acid, a renewable resource, in accordance with green chemistry principles. This supports sustainable practices in industries that use sodium acetate trihydrate in their processes.
Current research is exploring new applications and improving the efficiency of sodium acetate trihydrate production. Innovations in green chemistry and materials science will lead to new uses in energy storage, advanced materials and environmental technologies, expanding its role in various fields.
References
2021. ZnO nanorods/Fe3O4-graphene oxide/metal-organic framework nanocomposite: recyclable and robust photocatalyst for degradation of pharmaceutical pollutants. *Environmental science and pollution research international*, 28(18). DOI: 10.1007/s11356-020-12253-2
2008. Preservation of ancient DNA in thermally damaged archaeological bone. *Die Naturwissenschaften*, 96(2). DOI: 10.1007/s00114-008-0478-5
2001. Amino Acid Analysis Using Precolumn Derivatization with 6-Aminoquinolyl-N-Hydroxysuccinimidyl Carbamate. *Methods in molecular biology (Clifton, N.J.)*, 159. DOI: 10.1385/1-59259-047-0:039
|