Online Database of Chemicals from Around the World
| Insight Finechem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ifc.cn | |||
 | +86 (10) 8278-0291 | |||
 | +86 (10) 8278-0289 | |||
 | jane@ifc.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Beijing TND Science and Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tndtech.com.cn | |||
 | +86 (10) 8075-1706 +86 13521671946 | |||
 | +86 (10) 8075-1706 | |||
 | sales@tndtech.com.cn | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Lift Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.liftchem.com | |||
 | +86 (22) 8834-5196 | |||
 | sales@liftchem.com | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Nivon Specialties | India | |||
|---|---|---|---|---|
 | www.navoneindia.com | |||
 | +91 9323789882 | |||
 | nivonpharma@yahoo.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Other amino acid derivatives |
|---|---|
| Name | 4-Dimethylaminobenzoic acid |
| Synonyms | p-(N,N'-Dimethylamino) benzoic acid |
| Molecular Structure | 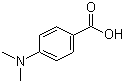 |
| Molecular Formula | C9H11NO2 |
| Molecular Weight | 165.19 |
| Protein Sequence | X |
| CAS Registry Number | 619-84-1 |
| EC Number | 210-615-8 |
| SMILES | CN(C)C1=CC=C(C=C1)C(=O)O |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 241 - 243 °C (Decomposes) (Expl.) |
| Boiling point | 315.8±25.0 °C 760 mmHg (Calc.)* |
| Flash point | 144.8±23.2 °C (Calc.)*, 170 °C (Expl.) |
| Index of refraction | 1.594 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Dimethylaminobenzoic acid is an aromatic carboxylic acid compound with the molecular formula C9H11NO2. It consists of a benzoic acid core substituted at the para (4-) position with a dimethylamino group (–N(CH3)2). This structural arrangement gives the compound both acidic (carboxylic acid) and basic (amine) functional properties, and it is commonly used in organic synthesis, pharmaceutical development, and analytical chemistry. The compound is a derivative of benzoic acid and was first synthesized during early 20th-century investigations into substituted aromatic acids and amines. It is typically prepared by dimethylation of 4-aminobenzoic acid using methylating agents such as dimethyl sulfate or methyl iodide under basic conditions. 4-Dimethylaminobenzoic acid has been used in various synthetic pathways as a starting material or intermediate. The electron-donating nature of the dimethylamino group influences the reactivity of the aromatic ring, making it more reactive toward electrophilic substitution and useful in the formation of dyes, pH indicators, and functionalized materials. In particular, it is a known intermediate in the synthesis of azo dyes and fluorescent compounds, where its electron-rich structure enhances chromophore behavior. In pharmaceutical chemistry, derivatives of 4-dimethylaminobenzoic acid have been explored for their bioactivity and drug-like properties. Some esters of this compound, such as its ethyl ester, are used as UV absorbers in sunscreen formulations and as excipients in topical medications. Related compounds have also been studied for local anesthetic activity, based on their structural similarity to para-aminobenzoic acid (PABA), a known pharmacophore in anesthetic agents. The compound has also served a role in analytical chemistry, particularly in spectrophotometric and chromatographic methods. Its conjugated aromatic system and the auxochromic dimethylamino group contribute to its UV-visible absorbance properties, which are useful for detection and quantification in chemical assays. 4-Dimethylaminobenzoic acid is a solid at room temperature, typically appearing as a light yellow crystalline powder. It is sparingly soluble in water but more soluble in alcohols and organic solvents like ethanol or acetone. It has a melting point in the range of 170–175 °C. The carboxylic acid group can be readily converted into salts, esters, or amides, allowing for further modification and incorporation into complex molecules. Safety data indicate that the compound should be handled with care. Although it has relatively low acute toxicity, it may cause irritation to the skin, eyes, or respiratory tract upon direct exposure. Proper laboratory safety practices, including gloves and eye protection, are recommended during use. In summary, 4-dimethylaminobenzoic acid is a synthetically useful aromatic acid known for its role in dye chemistry, UV-absorbing agents, and pharmaceutical intermediates. Its electron-rich, dual-functional structure allows it to participate in diverse chemical transformations relevant to industrial and academic research. References 2017. Studies on the formation of formaldehyde during 2-ethylhexyl 4-(dimethylamino)benzoate demethylation in the presence of reactive oxygen and chlorine species. Environmental science and pollution research international, 24(10). DOI: 10.1007/s11356-017-8477-8 2014. Evaluation of benzoic acid derivatives as sirtuin inhibitors. Bioorganic & Medicinal Chemistry Letters, 24(1). DOI: 10.1016/j.bmcl.2013.11.004 1981. Slow-acting Amine Polymerization Accelerators. Para-dimethylaminobenzoic Acid and Its Ethyl Ester. Journal of Dental Research, 60(7). DOI: 10.1177/00220345810600070701 |
| Market Analysis Reports |