Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Changzhou Carbochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.carbo-chem.com | |||
 | +86 (519) 8918-1862 +86 13775204319 | |||
 | +86 (519) 8918-1862 | |||
 | gao@carbo-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Dishman Pharmaceuticals & Chemicals Limited | USA | |||
|---|---|---|---|---|
 | www.dishmangroup.com | |||
 | +1 (732) 560-4300 | |||
 | +1 (732) 560-4343 | |||
 | bhaveshoza@dishmangroup.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Joyochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.joyochem.com | |||
 | +86 13290333633 | |||
 | sales@joyochem.com | |||
 | QQ Chat | |||
 | WeChat: 13290333633 | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Pure Chemistry Scientific Inc | USA | |||
|---|---|---|---|---|
 | www.purechems.com | |||
 | +1 (888) 588-9418 | |||
 | +1 (617) 206-9595 | |||
 | sales@purechems.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shenzhen Dieckmann Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dkmchem.com | |||
 | +86 (755) 8921-3952 | |||
 | +86 (755) 8481-3563 | |||
 | sale@dkmchem.com | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Sunsure Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunsurechem.com | |||
 | +86 (25) 5853-5435 | |||
 | +86 (25) 5853-5435 | |||
 | sales@sunsurechem.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Endotherm GmbH | Germany | |||
|---|---|---|---|---|
 | www.endotherm-lsm.com | |||
 | +49 (681) 3946-7570 | |||
 | +49 (681) 3946-7571 | |||
 | info@endotherm.de | |||
| Chemical manufacturer | ||||
| Classification | API >> Vitamins and minerals >> Vitamin AD drugs |
|---|---|
| Name | Calcifediol |
| Synonyms | (5Z,7E)-9,10-Secocholesta-5,7,10(19)-triene-3b,25-diol monohydrate; 25-Hydroxycholecalciferol monohydrate; 25-OHD3; 25-Hydroxyvitamin D3 monohydrate |
| Molecular Structure | 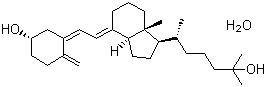 |
| Molecular Formula | C27H44O2.H2O |
| Molecular Weight | 418.65 |
| CAS Registry Number | 63283-36-3 |
| EC Number | 621-370-5 |
| SMILES | C[C@H](CCCC(C)(C)O)[C@H]1CC[C@@H]2[C@@]1(CCC/C2=CC=C/3C[C@H](CCC3=C)O)C.O |
| Solubility | DMSO 72 mg/mL, Water 70 mg/mL (Expl.) |
|---|---|
| Hazard Symbols |   GHS06;GHS08 Danger Details GHS06;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H301-H311-H330-H372 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P262-P264-P270-P271-P280-P284-P301+P316-P302+P352-P304+P340-P316-P319-P320-P321-P330-P361+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Calcifediol, with the molecular formula C27H44O2 and CAS number 19356-17-3, is a naturally occurring vitamin D metabolite, also known as 25-hydroxyvitamin D3 or 25(OH)D3. It is the primary circulating form of vitamin D in the human body, serving as a biomarker for vitamin D status and a precursor to the active hormone calcitriol. Its discovery and applications are well-documented in the literature, rooted in the study of vitamin D metabolism and its role in calcium homeostasis and bone health. The discovery of calcifediol emerged from research in the mid-20th century to elucidate the metabolic pathway of vitamin D. In the 1960s, scientists, including Hector DeLuca and colleagues at the University of Wisconsin, identified calcifediol as the major circulating metabolite of cholecalciferol (vitamin D3), formed by hepatic hydroxylation. Using radiolabeled vitamin D3, they demonstrated that the liver enzyme 25-hydroxylase (CYP2R1) converts cholecalciferol to 25-hydroxyvitamin D3, which was isolated and characterized by 1968. This breakthrough clarified the two-step activation process of vitamin D, with calcifediol as the intermediate that is further hydroxylated in the kidneys to calcitriol (1,25-dihydroxyvitamin D3). The discovery addressed the need to understand vitamin D’s role in preventing rickets, osteoporosis, and other metabolic disorders, building on advances in chromatography, mass spectrometry, and enzymatic studies. Calcifediol’s identification led to its use in clinical diagnostics and therapeutics by the 1970s. Synthetically, calcifediol is produced from cholecalciferol, which is derived from 7-dehydrocholesterol extracted from lanolin or fish oil. The key step involves enzymatic or chemical 25-hydroxylation. In industrial processes, cholecalciferol is subjected to biomimetic hydroxylation using cytochrome P450 enzymes or chemical oxidation with reagents like selenium dioxide or manganese oxide, selectively introducing the hydroxyl group at the 25-position of the side chain. The reaction is followed by purification via high-performance liquid chromatography to yield calcifediol as a white crystalline powder. Alternatively, calcifediol can be isolated from animal liver extracts, but synthetic methods are preferred for scalability and purity. These processes rely on established protocols in steroid chemistry and enzymatic catalysis, ensuring high stereospecificity and yield. The primary application of calcifediol is in clinical medicine for the treatment and prevention of vitamin D deficiency and related disorders. As a therapeutic agent, it is used to manage conditions such as osteoporosis, osteomalacia, hypoparathyroidism, and chronic kidney disease, where impaired renal 1-hydroxylation limits calcitriol production. Calcifediol is administered orally or via injection, offering advantages over cholecalciferol due to its higher potency and faster onset of action, as it bypasses hepatic 25-hydroxylation. It raises serum 25-hydroxyvitamin D levels more effectively, supporting calcium absorption and bone mineralization. In diagnostics, calcifediol levels in blood are measured via immunoassays or liquid chromatography-tandem mass spectrometry to assess vitamin D status, with levels below 20 ng/mL indicating deficiency. It is also used in research to study vitamin D metabolism, receptor interactions, and non-skeletal effects, including immune modulation and cancer prevention. In academic research, calcifediol is investigated for its role in the vitamin D endocrine system, particularly the regulation of 25-hydroxylase and 1-alpha-hydroxylase enzymes. Its pharmacokinetics and binding to vitamin D-binding protein are studied to optimize dosing regimens. The compound’s significance lies in its dual role as a therapeutic agent and diagnostic marker, reflecting progress in vitamin D research and steroid biochemistry. By enabling effective management of vitamin D-related disorders, calcifediol has become a cornerstone in endocrinology and clinical nutrition. References 2024. Efficacy and Safety of Weekly Calcifediol Formulations (75 and 100 µg) in Subjects with Vitamin D Deficiency: A Phase II/III Randomised Trial. Nutrients, 16(22). DOI: 10.3390/nu16223796 2024. Native vitamin D in CKD and renal transplantation: meaning and rationale for its supplementation. Journal of Nephrology, 37(7). DOI: 10.1007/s40620-024-02055-x 1987. Hyperparathyroidism and Low Serum Osteocalcin Despite Vitamin D Replacement in Primary Biliary Cirrhosis. The Journal of Clinical Endocrinology and Metabolism, 64(5). DOI: 10.1210/jcem-64-5-873 |
| Market Analysis Reports |