Online Database of Chemicals from Around the World
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Pyrimidine |
|---|---|
| Name | 2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine |
| Molecular Structure | 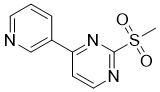 |
| Molecular Formula | C10H9N3O2S |
| Molecular Weight | 235.26 |
| CAS Registry Number | 637354-25-7 |
| SMILES | CS(=O)(=O)C1=NC=CC(=N1)C2=CN=CC=C2 |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.576, Calc.* |
| Boiling Point | 486.3±37.0 °C (760 mmHg), Calc.* |
| Flash Point | 247.9±26.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details |
| SDS | Available |
|
2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine is a notable chemical compound recognized for its unique structure and diverse applications in scientific research. This molecule features a pyrimidine ring system substituted with a methylsulfonyl group and a pyridinyl group, which together influence its chemical reactivity and potential utility. The discovery of 2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine emerged from efforts to develop functionalized pyrimidine derivatives. The pyrimidine ring, a six-membered heterocycle containing nitrogen, is a key structure in various biologically active compounds. The addition of a methylsulfonyl group at position 2 and a pyridinyl group at position 4 introduces distinct electronic and steric properties, enhancing the molecule’s versatility. In medicinal chemistry, 2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine has attracted attention for its potential therapeutic applications. The pyrimidine core is associated with numerous bioactive compounds, and the specific substituents in this molecule can modulate its interaction with biological targets. Researchers are exploring how the methylsulfonyl and pyridinyl groups affect the compound’s biological activity, aiming to identify new drug candidates with enhanced efficacy. For example, such modifications can influence the molecule’s ability to inhibit specific enzymes or interact with receptors, making it a candidate for further development in drug discovery. The compound also has applications in materials science and organic synthesis. Its structure allows it to function as an intermediate in the synthesis of more complex molecules. The methylsulfonyl and pyridinyl groups enable it to participate in a variety of chemical reactions, facilitating the development of new organic materials and specialty chemicals. This includes applications in the creation of organic electronics, coatings, and other advanced materials where its unique chemical properties can be leveraged. Furthermore, 2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine is valuable in research exploring new chemical reactivity patterns. Its ability to engage in diverse chemical transformations makes it a useful tool for chemists seeking to design and synthesize novel compounds. The combination of the methylsulfonyl and pyridinyl groups introduces reactivity that can be exploited in various synthetic applications. In summary, 2-(Methylsulfonyl)-4-(pyridin-3-yl)pyrimidine is a compound of significant interest due to its unique structure and reactivity. Its potential applications in medicinal chemistry, materials science, and organic synthesis underscore its importance in advancing research and development across multiple fields. References 2010. A novel synthesis of imatinib and its intermediates. Monatshefte für Chemie - Chemical Monthly, 141(7). DOI: 10.1007/s00706-010-0334-0 2011. Bis{3-[2-(methyl-sulfonyl)pyrimidin-4-yl]pyridinium} tetra-chloridocadmium. Acta crystallographica. Section E, Structure reports online, 67(12). DOI: 3238717 2012. CCDC 705138: Experimental Crystal Structure Determination. DOI: 10.5517/ccrnrdj |
| Market Analysis Reports |