Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| La Mesta Chimie Fine S.A.S. | France | |||
|---|---|---|---|---|
 | www.la-mesta.com | |||
 | +33 (4) 9208-5300 | |||
 | +33 (4) 9208-5340 | |||
 | info@la-mesta.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Taixing Joxin Bio-tec Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.joxbio.com | |||
 | +86 (523) 8755-8858 | |||
 | +86 (523) 8755-8858 / 8761-2088 | |||
 | yhj@joxbio.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Classification | Chemical reagent >> Organic reagent >> Nitro compound |
|---|---|
| Name | 3-Nitrophthalic anhydride |
| Synonyms | 4-nitro-2-benzofuran-1,3-dione |
| Molecular Structure | 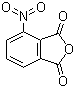 |
| Molecular Formula | C8H3NO5 |
| Molecular Weight | 193.11 |
| CAS Registry Number | 641-70-3 |
| EC Number | 211-373-6 |
| SMILES | C1=CC2=C(C(=C1)[N+](=O)[O-])C(=O)OC2=O |
| Density | 1.7±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 163-165 °C (Expl.) |
| Index of Refraction | 1.660, Calc.* |
| Boiling Point | 411.8±28.0 °C (760 mmHg), Calc.* |
| Flash Point | 227.5±26.0 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
3-Nitrophthalic anhydride is a chemical compound with a molecular formula of C8H5NO4, commonly used in various industrial applications, particularly in the production of dyes, plastics, and pharmaceuticals. This compound belongs to the class of phthalic anhydrides, which are derivatives of phthalic acid. The presence of a nitro group at the 3-position of the aromatic ring distinguishes it from other phthalic anhydride compounds, contributing to its unique chemical properties and reactivity. The discovery of 3-nitrophthalic anhydride can be traced back to the early 20th century when researchers were investigating the various derivatives of phthalic acid. The nitration of phthalic anhydride to introduce a nitro group at the 3-position resulted in the formation of this compound. Over time, it gained attention for its ability to participate in various chemical reactions, making it useful in a variety of chemical processes and applications. One of the primary applications of 3-nitrophthalic anhydride is in the production of specialty dyes and pigments. The presence of the nitro group in the molecule enhances the compound's ability to interact with various substrates, enabling the synthesis of colorants with desirable properties. This makes 3-nitrophthalic anhydride a valuable intermediate in the manufacture of azo dyes, which are widely used in textiles, inks, and coatings. The compound's ability to undergo further chemical modifications allows for the development of a wide range of dyes with varying shades and applications. In addition to its role in dye production, 3-nitrophthalic anhydride is also utilized in the synthesis of high-performance polymers and plastics. Its chemical structure allows it to participate in the formation of polyesters and polyamides, which are materials used in various industries, including automotive, electronics, and construction. The incorporation of the nitro group in the polymer backbone can enhance certain properties of the material, such as thermal stability, mechanical strength, and chemical resistance, making it suitable for use in demanding applications. Pharmaceutical applications of 3-nitrophthalic anhydride also exist, particularly in the synthesis of certain active pharmaceutical ingredients (APIs). The compound can be used as an intermediate in the production of drugs that require specific chemical functional groups for their biological activity. For example, the nitro group in 3-nitrophthalic anhydride can serve as a key component in the synthesis of drugs that target specific enzymes or receptors in the body, contributing to the development of new therapeutic agents. Furthermore, 3-nitrophthalic anhydride plays a role in the field of organic synthesis as a reagent in various reactions. It is commonly used in the preparation of other functionalized aromatic compounds, which can then be utilized in the synthesis of more complex molecules. The compound's reactivity makes it a valuable tool in laboratories and industrial settings where the production of specialty chemicals is required. Despite its utility in various applications, 3-nitrophthalic anhydride must be handled with care due to its potential to cause irritation to the skin, eyes, and respiratory system. Proper safety measures, including protective equipment and ventilation, should be observed when working with this compound to avoid exposure. Additionally, as with other nitroaromatic compounds, 3-nitrophthalic anhydride should be managed in accordance with environmental regulations to prevent contamination and ensure safe disposal. In conclusion, 3-nitrophthalic anhydride is a versatile chemical compound with a wide range of applications in industries such as dye manufacturing, polymer production, pharmaceuticals, and organic synthesis. Its unique chemical structure, which includes a nitro group at the 3-position of the aromatic ring, makes it an important intermediate in the synthesis of various products. Continued research into its properties and potential uses may expand its applications further, especially in fields that require specialized chemical reactivity and functionalization. References 2008. Analytical method for determination of allylic isoprenols in rat tissues by liquid chromatography/tandem mass spectrometry following chemical derivatization with 3-nitrophtalic anhydride. Journal of Pharmaceutical and Biomedical Analysis, 47(4-5). DOI: 10.1016/j.jpba.2008.01.032 2013. Facile synthesis of 3-substituted quinazoline-2,4-dione and 2,3-di-substituted quinazolinone derivatives. Chemical Papers, 67(1). DOI: 10.2478/s11696-012-0266-8 1994. Sensitive enantiomeric separation of aliphatic and aromatic amines using aromatic anhydrides as non-chiral derivatizing agents. Journal of chromatography. A, 664(2). DOI: 10.1016/0021-9673(94)80410-9 |
| Market Analysis Reports |