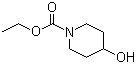
Ethyl 4-hydroxypiperidine-1-carboxylate is an organic compound belonging to the class of carbamate derivatives, characterized by a piperidine ring substituted at the 4-position with a hydroxyl group and bearing an ethyl carbamate moiety at the nitrogen atom of the ring. Its molecular formula is C8H15NO3. The compound is of interest in organic synthesis as an intermediate and building block for the preparation of various pharmaceuticals and bioactive molecules.
The compound is typically synthesized via the reaction of 4-hydroxypiperidine with ethyl chloroformate or ethyl carbonochloridate, which introduces the ethyl carbamate protecting group at the nitrogen atom. This modification stabilizes the piperidine ring nitrogen and allows for selective functionalization of other positions on the ring.
Ethyl 4-hydroxypiperidine-1-carboxylate is commonly used in medicinal chemistry as a versatile intermediate for the synthesis of compounds with potential biological activity, including central nervous system agents, analgesics, and anti-inflammatory drugs. The hydroxyl group at the 4-position can be further derivatized to introduce various substituents, allowing the generation of structural diversity necessary for drug discovery.
In synthetic applications, the compound serves as a precursor in the construction of heterocyclic compounds through nucleophilic substitution, oxidation, and other organic transformations. The carbamate protecting group is stable under many reaction conditions but can be selectively removed to yield the free amine when desired.
Analytical characterization of ethyl 4-hydroxypiperidine-1-carboxylate is performed using nuclear magnetic resonance (NMR) spectroscopy, infrared (IR) spectroscopy, and mass spectrometry (MS). These techniques confirm the presence of the carbamate group, hydroxyl functionality, and overall molecular structure. Purity and identity are critical for its application in pharmaceutical synthesis.
Physically, the compound is generally a colorless to pale liquid or solid depending on purity and preparation method. It is soluble in common organic solvents such as ethanol, dichloromethane, and acetone, facilitating its use in synthetic protocols.
In summary, ethyl 4-hydroxypiperidine-1-carboxylate is a carbamate derivative of 4-hydroxypiperidine widely utilized as a chemical intermediate in pharmaceutical synthesis. Its functional groups allow for diverse chemical modifications, making it valuable in the preparation of various biologically active compounds.
References
none
|




































 GHS07 Warning Details
GHS07 Warning Details