Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Shaanxi Scidoor Hi-Tech Biology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.scidoor.com | |||
 | +86 (29) 6296-0300 6296-0309 6296-0305 | |||
 | +86 (29) 6296-0320 | |||
 | sales@scidoor.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Extrasynthese Chemical S.A.S. | France | |||
|---|---|---|---|---|
 | www.extrasynthese.com | |||
 | +33 (47) 898-2034 | |||
 | +33 (47) 898-1945 | |||
 | info@extrasynthese.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Tetrahedron Scientific Inc. | China | |||
|---|---|---|---|---|
 | www.thsci.com | |||
 | +86 (571) 8848-9473 +86 18072878644 | |||
 | +86 (571) 8848-9473 | |||
 | sales@thsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Orient Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.greenchemintl.com | |||
 | +86 (21) 5996-9669 | |||
 | +86 (21) 5996-9200 | |||
 | jackson@greenchemintl.com info@greenchemintl.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Yangzhou Rixing Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rxbiochem.com | |||
 | +86 (514) 8095-5801 | |||
 | clint@rxgp.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Nanjing Healthherb Bio-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.healthherb.cn | |||
 | +86 (25) 8663-8485 | |||
 | +86 (25) 8331-8076 | |||
 | 2665774077@qq.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2014 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Creative Proteomics | USA | |||
|---|---|---|---|---|
 | www.creative-proteomics.com | |||
 | +1 (631) 593-0501 | |||
 | +1 (631) 614-7828 | |||
 | contact@creative-proteomics.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shandong Caman Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cmpharm.com.cn | |||
 | +86 (0539) 710-9900 | |||
 | 1215459684@qq.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Qingdao Wanyuan Mountain Biontech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bionutrichem.com | |||
 | +86 15092119928 | |||
 | cuiningning@bionutrichem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Maanshan Tiantai Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tiantaibio-tech.cn | |||
 | +86 (0555) 888-9890 | |||
 | +86 (0555) 888-9890 | |||
 | shenguangguang@tiantaibio-tech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Aajiang Jiuzhou Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuzhou-chem.com | |||
 | +86 13454675544 | |||
 | jamie@jiuzhou-chem.com | |||
 | QQ Chat | |||
 | WeChat: +86 18632988332 | |||
 | WhatsApp:+86 18632988332 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Zhejiang Hisun Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hisunpharm.com | |||
 | +86 13651899693 | |||
 | yiwen.sun@hisunpharm.com | |||
| Chemical manufacturer since 1956 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Orgentis Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.orgentis.com | |||
 | +49 39482-355 | |||
 | +49 39482-79071 | |||
 | sales@orgentis.com | |||
| Chemical manufacturer since 1991 | ||||
| Omicron Biochemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.omicronbio.com | |||
 | +1 (574) 287-6910 | |||
 | +1 (574) 287-7165 | |||
 | omicron@omicronbio.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Yantai Dongcheng Biochemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dcb-group.com | |||
 | +86 (535) 637-3853 637-8569 637-0505 | |||
 | +86 (535) 637-3885 | |||
 | sales@dcb-group.com | |||
| Chemical manufacturer since 1998 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Classification | API >> Antipyretic analgesics >> Anti-gout medicine |
|---|---|
| Name | D-Glucosamine hydrochloride |
| Synonyms | 2-Amino-2-deoxy-D-glucopyranose hydrochloride |
| Molecular Structure | 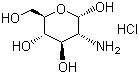 |
| Molecular Formula | C6H13NO5.HCl |
| Molecular Weight | 215.63 |
| CAS Registry Number | 66-84-2 |
| EC Number | 200-638-1 |
| SMILES | C([C@H]([C@H]([C@@H]([C@H](C=O)N)O)O)O)O.Cl |
| Melting point | 190 - 194 °C (Decomposes) (Expl.) |
|---|---|
| alpha | 72.5 ° (c=2, H2O, 5hrs.) |
| Water solubility | soluble |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
D-Glucosamine hydrochloride is the hydrochloride salt of D-glucosamine, a naturally occurring amino sugar derived from the hydrolysis of chitin, the structural component found in the exoskeletons of crustaceans and insects, as well as in fungal cell walls. D-Glucosamine itself is an essential precursor in the biosynthesis of glycosaminoglycans, which are critical constituents of cartilage, skin, tendons, and ligaments. The hydrochloride form enhances the stability and solubility of glucosamine, making it more suitable for pharmaceutical and nutraceutical applications. The discovery of D-glucosamine dates back to the late 19th century when Georg Ledderhose first isolated the compound in 1876 through the hydrolysis of chitin with concentrated hydrochloric acid. This process yielded D-glucosamine hydrochloride as a stable crystalline product. Early studies characterized the compound’s structure through careful chemical degradation and analysis, ultimately confirming the presence of an amino group at the 2-position of the glucose backbone. D-Glucosamine hydrochloride has been widely studied for its biological significance and applications. It plays a vital role in the biosynthetic pathway leading to the formation of important macromolecules such as hyaluronic acid, keratan sulfate, and heparan sulfate. These glycosaminoglycans are critical for maintaining the structural integrity and function of connective tissues. In the human body, glucosamine is synthesized from fructose-6-phosphate via the enzyme glutamine-fructose-6-phosphate amidotransferase, indicating its endogenous importance. The compound has gained prominence for its application in the management of osteoarthritis, a degenerative joint disease characterized by cartilage breakdown and inflammation. D-Glucosamine hydrochloride is widely marketed as a dietary supplement intended to support joint health. Numerous clinical studies have investigated its efficacy, with some reporting that supplementation can alleviate joint pain and improve mobility, although results vary depending on study design and patient population. It is generally considered to have a favorable safety profile, with mild gastrointestinal discomfort being the most commonly reported side effect. In addition to its use in joint health, D-glucosamine hydrochloride serves as a starting material for the synthesis of a variety of biologically active compounds. It is a key intermediate in the production of N-acetyl-D-glucosamine, which has applications in cosmetics and dermatology due to its moisturizing and anti-aging properties. Furthermore, derivatives of glucosamine have been explored for their potential antimicrobial, antitumor, and anti-inflammatory activities, reflecting the versatility of this molecule in medicinal chemistry. Industrial production of D-glucosamine hydrochloride typically involves the chemical hydrolysis of chitin under acidic conditions, followed by neutralization and crystallization to obtain the pure salt. Alternative biotechnological methods employing enzymatic hydrolysis have also been developed to improve efficiency and reduce environmental impact. Quality control during production is critical, as impurities such as heavy metals or residual proteins must be minimized to meet pharmaceutical-grade standards. The compound’s chemical properties, including its high solubility in water and its reactive amino group, make it a useful reagent in carbohydrate chemistry. It can undergo various chemical modifications, such as acetylation, sulfation, and conjugation with other biomolecules, to create novel materials for biomedical applications, including drug delivery systems and tissue engineering scaffolds. D-Glucosamine hydrochloride’s stability as a salt also facilitates its formulation into tablets, capsules, and powders for oral administration. Its compatibility with other commonly used joint supplements, such as chondroitin sulfate and methylsulfonylmethane (MSM), has led to the development of combination products aimed at enhancing therapeutic outcomes for individuals with joint disorders. References 1973. Anomeric effects in Schiff bases of D-glucosamine with aromatic aldehydes. Journal of Applied Spectroscopy, 19(6). DOI: 10.1007/bf00611086 2001. The Amadori and Heyns Rearrangements: Landmarks in the History of Carbohydrate Chemistry or Unrecognized Synthetic Opportunities? Topics in Current Chemistry, 215. DOI: 10.1007/3-540-44422-x_6 2004. Surface properties of surfactants derived from natural products. Part 1: Syntheses and structure/property relationships—Solubility and emulsification. Journal of Surfactants and Detergents, 7(2). DOI: 10.1007/s11743-004-0298-6 |
| Market Analysis Reports |