Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Fenhe Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fenhechem.com | |||
 | +86 (021) 3392-6068 | |||
 | julius.wei@fenhechem.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Parkway Scientific | USA | |||
|---|---|---|---|---|
 | www.parkwayscientific.com | |||
 | +1 (336) 587-2898 | |||
 | +1 (707) 215-0888 | |||
 | sales@parkwayscientific.com | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Aminopyrimidine |
|---|---|
| Name | 4-(3-Pyridinyl)-2-aminopyrimidine |
| Synonyms | 4-(3-Pyridinyl)-2-pyrimidinamine |
| Molecular Structure | 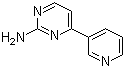 |
| Molecular Formula | C9H8N4 |
| Molecular Weight | 172.19 |
| CAS Registry Number | 66521-66-2 |
| EC Number | 835-883-9 |
| SMILES | C1=CC(=CN=C1)C2=NC(=NC=C2)N |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
4-(3-Pyridinyl)-2-aminopyrimidine is an organic compound with the molecular formula C9H8N4. This compound features a pyrimidine ring with two substituents: an amino group (-NH2) at the 2-position and a pyridine ring at the 4-position. The unique combination of these two heterocyclic rings imparts distinctive chemical properties and biological activities to the compound, making it a valuable subject of study in medicinal chemistry and drug development. The discovery of 4-(3-Pyridinyl)-2-aminopyrimidine is part of the broader exploration of pyrimidine derivatives, which are known for their diverse biological activities and applications. Pyrimidines are six-membered aromatic rings containing nitrogen atoms, and their derivatives have been extensively studied for their potential as pharmaceutical agents. The introduction of the pyridine ring into the pyrimidine structure creates a compound with enhanced chemical versatility and potential therapeutic properties. One of the primary applications of 4-(3-Pyridinyl)-2-aminopyrimidine is in drug development. This compound serves as a key intermediate in the synthesis of various pharmaceutical agents. Its structure allows it to interact with biological targets, such as enzymes or receptors, due to the presence of the amino group and the pyridine ring. These interactions can lead to the modulation of biological processes, making 4-(3-Pyridinyl)-2-aminopyrimidine a candidate for developing drugs that target specific pathways or conditions. For instance, 4-(3-Pyridinyl)-2-aminopyrimidine has been investigated for its potential as an anti-cancer agent. The compound's ability to inhibit certain kinases—enzymes involved in cell signaling and growth—has been explored in cancer research. Inhibition of these kinases can prevent the proliferation of cancer cells, making the compound a valuable starting point for the development of targeted cancer therapies. Additionally, this compound is studied for its potential in the treatment of other diseases. Its ability to interact with various biological targets suggests that it may have applications beyond oncology, including in the treatment of inflammatory diseases, infectious diseases, and neurological disorders. Researchers continue to investigate its efficacy and safety in preclinical and clinical trials to better understand its therapeutic potential. In the field of organic synthesis, 4-(3-Pyridinyl)-2-aminopyrimidine is used as a building block for creating more complex molecules. The functional groups on the pyrimidine ring can participate in further chemical reactions, allowing for the synthesis of derivatives with tailored properties. These derivatives can be explored for additional biological activities or used in the development of new materials with specific functionalities. Furthermore, 4-(3-Pyridinyl)-2-aminopyrimidine is employed in chemical research to study its reactivity and interaction with other molecules. Understanding these interactions can lead to the development of new synthetic methods and the design of compounds with desired properties. In summary, 4-(3-Pyridinyl)-2-aminopyrimidine is a significant compound in medicinal chemistry and organic synthesis. Its discovery and application have led to advancements in drug development, particularly in the search for targeted therapies for cancer and other diseases. The compound's unique structure and reactivity make it a valuable tool for researchers working to develop new pharmaceuticals and materials. References 2021. Inhibitors of Mycobacterium tuberculosis EgtD target both substrate binding sites to limit hercynine production. Scientific Reports, 11(1). DOI: 10.1038/s41598-021-01526-6 2016. A Practical Preparation of Imatinib Base. Synlett, 27(14). DOI: 10.1055/s-0035-1562498 2010. Design and synthesis of new anticancer pyrimidines with multiple-kinase inhibitory effect. Bioorganic & Medicinal Chemistry, 18(12). DOI: 10.1016/j.bmc.2010.04.037 |
| Market Analysis Reports |