Online Database of Chemicals from Around the World
| Vinati Organics Limited | India | |||
|---|---|---|---|---|
 | www.vinatiorganics.com | |||
 | +91 (22) 6124-0444 | |||
 | +91 (22) 2851-0729 | |||
 | siddhant.choudhary@vinatiorganics.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Shandong Goldym Group Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.goldym.com | |||
 | +86 (539) 621-3888 | |||
 | +86 (539) 621-2918 | |||
 | xieth2006@hotmail.com | |||
| Chemical manufacturer since 1975 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Puyang Wangda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wangdachem.com.cn | |||
 | +86 (393) 818-8309 | |||
 | +86 (393) 818-8306 | |||
 | wangdachem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| SACHEM, Inc. | USA | |||
|---|---|---|---|---|
 | www.sacheminc.com | |||
 | +1 (512) 421-4900 | |||
 | info@sacheminc.com | |||
| Chemical manufacturer since 1950 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Near Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.nearchemical.net | |||
 | +86 15615461505 | |||
 | belinda@nearchemical.net | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Weiyel Inc | China | |||
|---|---|---|---|---|
 | www.weiyel.com | |||
 | +44 7421898692 | |||
 | ivy@weiyelbncc.com | |||
 | QQ Chat | |||
 | WeChat: +8615672728568 | |||
 | WhatsApp:+44 7421898692 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Crystal Meths | UK | |||
|---|---|---|---|---|
 | crystalmeths.com | |||
 | 07365540947 | |||
 | mgrimaldi242@gmail.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2026 | ||||
| IRIS Biotech GmbH | Germany | |||
|---|---|---|---|---|
 | www.iris-biotech.de | |||
 | +49 (9231) 961-973 | |||
 | +49 (9231) 961-999 | |||
 | info@iris-biotech.de | |||
| Chemical manufacturer since 1788 | ||||
| Celanese Chemicals | USA | |||
|---|---|---|---|---|
 | www.celanese.com | |||
 | +1 (972) 443-4000 | |||
 | +1 (972) 443-2060 | |||
 | info@celanese.com | |||
| Chemical manufacturer | ||||
| Cambridge Isotope Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.isotope.com | |||
 | +1 (978) 749-8000 | |||
 | +1 (978) 749-2768 | |||
 | cilsales@isotope.com | |||
| Chemical manufacturer | ||||
| Thermo Fisher Scientific | USA | |||
|---|---|---|---|---|
 | www.fishersci.com | |||
 | +1 (800) 766-7000 | |||
 | +1 (800) 926-1166 | |||
 | fs.order@thermofisher.com | |||
| Chemical manufacturer | ||||
| Changshu Nanhu Industrial Chemical Factory | China | |||
|---|---|---|---|---|
 | www.csnh-chem.com | |||
 | +86 (512) 5244-4867 5188-2075 | |||
 | +86 (512) 5244-8121 | |||
 | zl19791220@126.com | |||
| Chemical manufacturer since 1993 | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Classification | Organic raw materials >> Alcohols, phenols, phenolic compounds and derivatives >> Acyclic alcohol |
|---|---|
| Name | Methanol |
| Synonyms | Carbinol; Methyl alcohol |
| Molecular Structure | 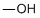 |
| Molecular Formula | CH3OH;CH4O |
| Molecular Weight | 32.04 |
| CAS Registry Number | 67-56-1 |
| EC Number | 200-659-6 |
| SMILES | CO |
| Solubility | miscible (water), soluble 20 mM (DMSO), 10 mM (ethanol) |
|---|---|
| Density | 0.7913 g/mL (20 °C) |
| Melting point | -98 °C |
| Boiling point | 64.7 °C |
| Refractive index | 1.3284 |
| Flash point | 12 °C |
| Hazard Symbols |    GHS02;GHS06;GHS08 Danger Details GHS02;GHS06;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H331-H311-H301-H370 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P262-P264-P270-P271-P280-P301+P316-P302+P352-P303+P361+P353-P304+P340-P308+P316-P316-P321-P330-P361+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1230 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Methanol, also known as methyl alcohol or wood alcohol, is the simplest alcohol with the chemical formula CH3OH. Its discovery dates back to the early 17th century when it was first isolated by the chemist Johann Rudolf Glauber in 1661. Glauber obtained methanol during the distillation of wood, a process known as destructive distillation, where organic materials are heated in the absence of air, leading to the breakdown of complex compounds into simpler ones. Commercial production of methanol began in the late 19th century, with the development of various synthesis methods, including the carbon monoxide hydrogenation process and the synthesis gas method, which combines hydrogen and carbon monoxide. The most significant advancement in methanol production occurred in the 1920s when German chemists developed the high-pressure catalytic process, allowing for large-scale synthesis and widespread use of methanol. Methanol has a diverse range of applications across various industries, making it one of the most important chemicals in the world. One of its primary uses is as a solvent and antifreeze agent due to its ability to lower the freezing point of water. Methanol is also an essential feedstock in the production of various chemicals, including formaldehyde, acetic acid, and various methyl esters. These chemicals serve as precursors in the manufacturing of plastics, resins, textiles, and paints. In the energy sector, methanol has gained attention as a clean-burning fuel alternative. It can be used as a fuel in internal combustion engines and fuel cells, providing a lower-emission option compared to traditional fossil fuels. Methanol can also be converted into dimethyl ether (DME), which can be used as a substitute for diesel fuel or as a clean cooking fuel. The pharmaceutical industry has recognized methanol's importance as a solvent in drug formulation and synthesis, enabling the production of various medicinal compounds. Furthermore, methanol is utilized in laboratories for analytical chemistry applications, including chromatography and spectrometry, due to its excellent solvent properties. However, despite its valuable applications, methanol poses significant health risks when improperly handled. It is highly toxic and can lead to methanol poisoning if ingested, inhaled, or absorbed through the skin. Symptoms of methanol poisoning include headache, dizziness, nausea, and, in severe cases, blindness or death. As a result, safety measures and regulations are crucial when using and handling methanol in both industrial and laboratory settings. In recent years, methanol has gained attention in the context of sustainability and environmental concerns. Researchers are exploring methods to produce methanol from renewable resources, such as biomass and carbon dioxide, which could contribute to reducing greenhouse gas emissions and dependence on fossil fuels. This potential for sustainable production aligns with global efforts to transition towards greener energy sources. In summary, methanol is a versatile chemical with a rich history, serving as a critical component in various industrial applications. Its importance spans from being a vital feedstock for chemical synthesis to a promising alternative fuel. As the world seeks more sustainable and cleaner energy sources, methanol's role is likely to expand, presenting new opportunities and challenges in its production and use. References 2025. Biomethanol production from renewable resources: a sustainable approach. Environmental science and pollution research international, 32(10). DOI: 10.1007/s11356-023-29616-0 2025. Unveiling the power of bene (Pistacia atlantica) hull scum: Boosting oxidative stability with methanolic extract and ferrous ions. Food Chemistry, 473. DOI: 10.1016/j.foodchem.2024.142142 2025. Polyvinylidene fluoride binder removal through subcritical methanol for efficient liberation of cathode materials from lithium-ion batteries. Waste management (New York, N.Y.), 191. DOI: 10.1016/j.wasman.2024.11.026 |
| Market Analysis Reports |